
You analyze a lot of biological samples. You deliver accurate, reliable data, consistently. You use sophisticated, high-end analytical techniques such as LC-MS/MS or GC-MS. And the pressure is always there to do it faster, with greater sensitivity and, if possible, at lower cost. Sample preparation can be critical for success.
Sample preparation enables more reliable identification and quantification of an analyte from a biological sample. The analyte is extracted (and often concentrated), from sample constituents that might interfere with the subsequent analysis. This can sound simple, but consider the basic challenges:
Choosing a sample preparation approach can have far-reaching consequences for your results as well as for the overall performance of your lab.
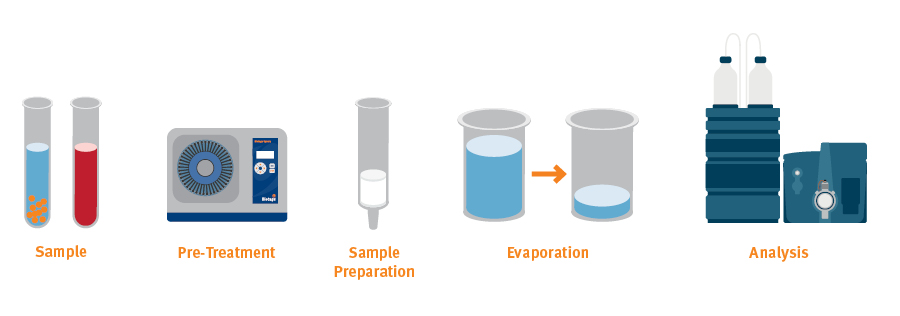
Fig. 1. An analytical workflow for LC-MS/MS or GC-MS can include several steps prior to the actual analysis.
Let’s take a look at the issues that you should consider and the options that are available for sample preparation.
This is a lengthy review packed with useful information about sample preparation. Download to read at your leisure !
Sample preparation is often considered a necessary inconvenience in the analytical process. You might want to disregard it as just extra work prior to the more glamorous analytical technique that delivers the result. Yet there are definite benefits you can gain from carefully chosen, appropriate, sample prep.
However, sample prep does add to the complexity of the analytical workflow and, unfortunately, there is no ‘one size fits all’. Extra steps take time and an incorrect choice of method can introduce errors rather than reducing them. So, what are your options and how do you choose between them?
There is a whole range of sample preparation techniques and each one has its place. Loosely, methods can be divided into ‘matrix scavenging’, designed to remove specific matrix components, and ‘targeted extraction’ where the characteristics of the analyte of interest directly impact the choice of sample prep technique and, frequently, components within the chosen technique.
Matrix Scavenging Techniques tend to be quicker, cheaper and simpler, whilst they usually require little or no method development compared with targeted extraction. However, they might not provide sufficiently high recovery or concentration to provide satisfactory detection limits.
Targeted Extraction Techniques, such as supported liquid extraction (SLE) and solid phase extraction (SPE) can be tailored more effectively to exploit analyte characteristics. They are often more complex, require more method development, and can be more expensive per analyzed sample. However, this is compensated for by delivering cleaner extracts with higher recoveries and potentially higher analytical sensitivity (lower detection limits).
These techniques are applicable to liquid samples as well as solid or semi-solid biological samples (such as tissue, hair or fingernails). For solid and semi-solid samples, additional sample pre-treatment is normally required to transfer analytes into a liquid phase before clean-up.
Before we look at more complicated approaches to sample preparation, let us consider the simplest. There are situations where the sample can be diluted then injected directly into, for example, an LC-MS/MS instrument. This is most often exploited with urine samples, which can be simply diluted 1:10 with water or buffer.
Clearly, there will be a direct negative impact on the limit of detection, but this might be acceptable. Furthermore, because all matrix components (such as salts, urea, proteins or phospholipids) are still present, they might reduce liquid chromatography (LC) column life, build up on the mass spectrometer (MS) source, or suppress ionization of the target analyte (if co-eluting with analyte from the liquid chromatography column).
If you can live with reduced sensitivity, afford the potential instrument downtime, accept costs for replacing LC columns more frequently, and accept having to regularly clean the MS source, then D&S is itself cheap, fast, and simple.
Another simple technique is to pass the sample through a 0.45 or 0.2 µm filter. Clearly, this will remove particulates that could clog an LC column, but won’t remove dissolved matrix components that can interfere in other ways. Commonly, filtration will be followed by dilution then injection. This method is also typically applied to urine although it may be appropriate for other biofluids. As with D&S, it is simple, quick and relatively cheap.
This is a matrix scavenging technique usually based on the familiar ‘acetonitrile crash’ approach. Simply add excess acetonitrile to the sample and either filter or centrifuge away the precipitated proteins.
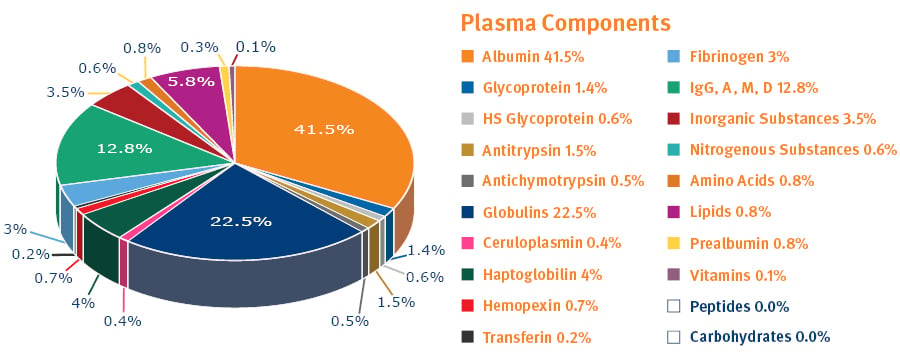
Fig. 2. Plasma is one protein-rich biological sample you might be analyzing1.
PPT is primarily used for blood, serum or plasma samples that carry a heavy protein load (see figure 2). It is available in a 96-well microtiter plate format where precipitation occurs in the wells and the clear protein-free samples are collected after passage through bottom filters (induced by pressure or vacuum) (see figure 3). This format is particularly suited to automated high-throughput assays, whereas using test tubes and a centrifuge is more appropriate for manual handling of small sample numbers.
PPT demands little method development, but it will not be suitable in all situations because it only removes proteins (not phospholipids) and doesn’t target any specific analyte. Nevertheless, the method is quick, simple and relatively inexpensive.
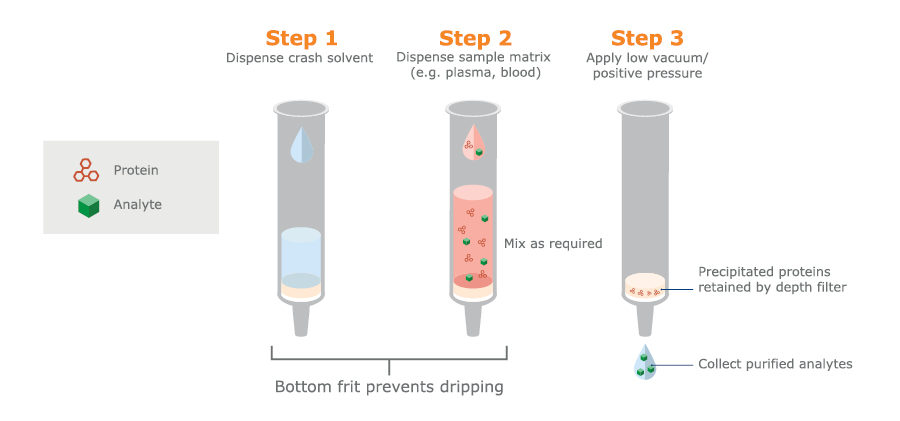
Fig. 3. Schematic PPT procedure used in 96-well microtiter plates with bottom filters.
Removal of phospholipids is extremely important in LC-MS/MS of biological fluids, particularly blood-based samples. Phospholipids are a family of compounds that can elute at various points throughout the chromatogram in LC and cause significant ion suppression in subsequent MS (see figure 4).
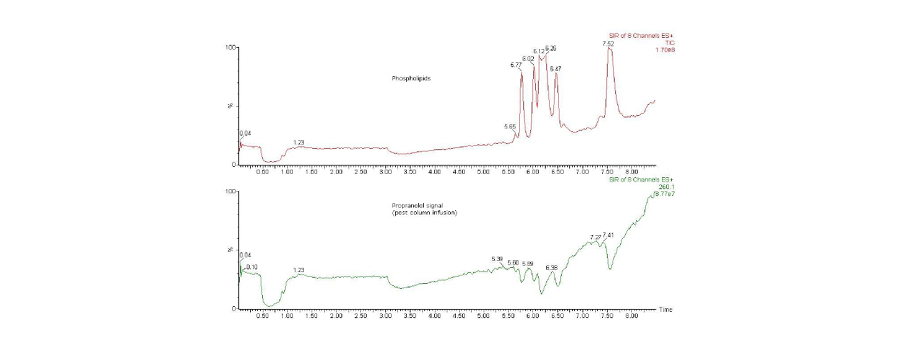
Fig. 4. Total ion chromatograms (TICs) from a post column infusion experiment. Top trace shows phospholipid species monitored by specific multiple reaction monitoring (MRM) transitions. Bottom trace shows a simultaneous fixed concentration flow of propranolol (specific MRM transition) added after the column, but before the MS source. Note that the signal decreases in the lower trace due to ion suppression by the phospholipid peaks.
Phospholipids can be conveniently removed following acetonitrile PPT using a scavenging adsorbent (see figure 5).
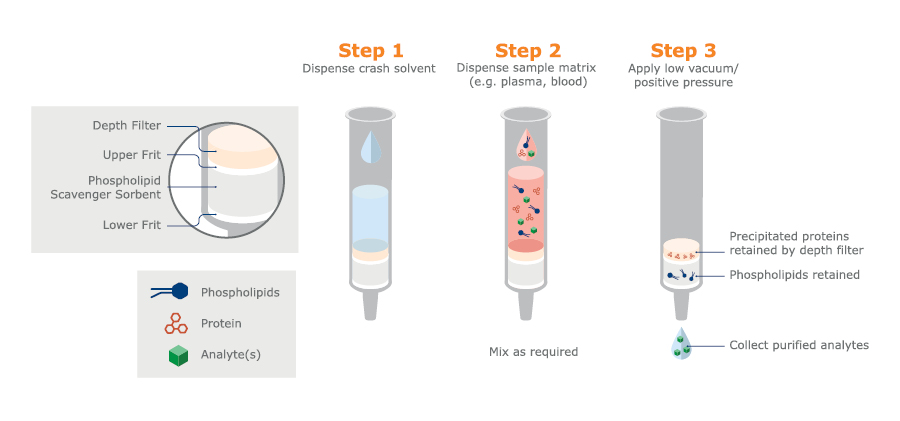
Fig. 5. Schematic procedure for PPT with isopropanol crash solvent, followed by PLD using a scavenging adsorbent. Available in column or 96-well microtiter plate formats.
PLD is another example of a matrix scavenging technique that does not target specific analytes. Although samples are rather clean after PLD, the technique might not be suitable for some kinds of analyses. However, method development is normally minimal, and PLD is simple and quick with relatively low costs.
This is a matrix scavenging technique designed principally for urine sample preparation. It is available commercially in column and 96-well plate formats. Matrix components such as urea, salts, pigments, creatinine, and proteins (residual enzyme from a hydrolysis step and possibly proteins from the urine itself), are removed by acetonitrile treatment followed by elution through two different sorbents. These steps can be performed in situ without any need for sample transfer (see figure 6).
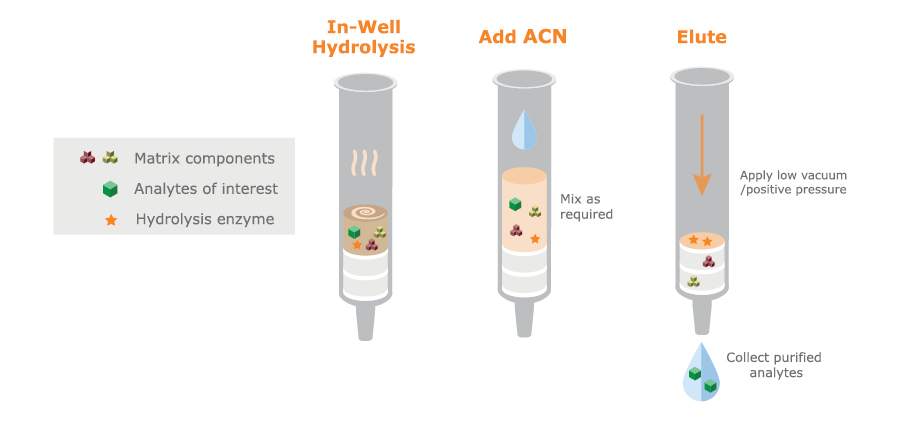
Fig. 6. Schematic DME procedure using in situ hydrolysis followed by acetonitrile precipitation and matrix removal by elution through separate scavenging and hygroscopic sorbents. Available in column or 96-well plate formats.
Urine Hydrolysis
In the body, most drugs are metabolized prior to excretion in urine or feces. Many drugs and metabolites are conjugated as a glucuronide or sulfate to increase water solubility and improve their elimination. Hydrolysis of urine specimens using a beta-glucuronidase (and/or sulfatase) converts such analytes to their ‘free’ form (de-conjugation) to increase analytical sensitivity, and improve chromatographic characteristics.
Urine sample hydrolysis involves incubating the sample (usually at elevated temperature) with an appropriate enzyme prior to cooling and quenching to stop the reaction.
The enzyme is abundant in living systems because it is an essential part of carbohydrate metabolism. Widely used laboratory sources of beta-glucuronidase include snails (Helix pomatia), abalone, and various recombinant forms.
Factors such as enzyme type and concentration, incubation temperature and time, and pH conditions will all affect hydrolysis efficiency, and different drug conjugates will hydrolyze at different rates. So, you will have to optimize hydrolysis conditions for your analytes of interest.
Once hydrolyzed, the sample will contain not only pre-existing matrix components (for urine these include salts, urea, creatinine, and pigments), but also excess enzyme, which can:
For these reasons, sample clean-up after hydrolysis is particularly important.
In conclusion, being a matrix scavenging technique, DME may not be suitable for certain analytes, but method development is minimal, and the method has a straightforward, pass-through workflow.
LLE is still widely used as a targeted sample preparation technique in bioanalysis, even if other approaches have partially replaced it. For low volume biofluid samples it can be performed in small vials, with manual shaking and use of a lab pipette to remove the unwanted layer – rather than using a separation funnel (which is easier for large samples such as drinking water or waste water). Large numbers of small-volume samples can be handled in a 96-well plate format, although it is challenging to do several steps in parallel.
LLE is effective and useful, particularly if you only have a few samples. It is also relatively cheap (if you don’t count labor costs). The issues come if you need to process large numbers of samples. Capping/shaking/settling of layers and pipetting can be challenging. It is difficult (although not impossible) to automate.
These issues have led to an increase in the popularity of alternative techniques, in particular supported liquid extraction (SLE), which works according to similar principles but is much easier to automate, avoids the risk of emulsion formation, and often provides a more efficient extraction process (i.e. with higher recovery).
Supported Liquid Extraction (SLE) is a targeted extraction technique for sample preparation before LC-MS/MS or GC-MS analysis. This extraction technique is based on the same chemistry as liquid-liquid extraction. However, instead of using a separation funnel and partitioning between two immiscible liquids – one aqueous and one organic – the extraction is performed on a solid support. Using SLE, an aqueous sample is loaded onto a 96-well plate or column containing an inert support (a form of diatomaceous earth). The aqueous sample forms a thin layer over the support to create a ‘stationary phase’.
The water-immiscible organic extraction solvent is then passed through the column/plate. As it passes over the thin layer of aqueous sample, analytes partition into the organic solvent, which is collected (see figure 7).
Clean-up is based on an analyte’s ability to partition into the water-immiscible organic extraction solvent. The method can be tailored to deliver high recovery of a specific analyte. Proteins, phospholipids, salts, pigments, etc., that might interfere with subsequent analyses tend not to be very soluble in the organic phase, so they are left behind in the aqueous phase on the column.
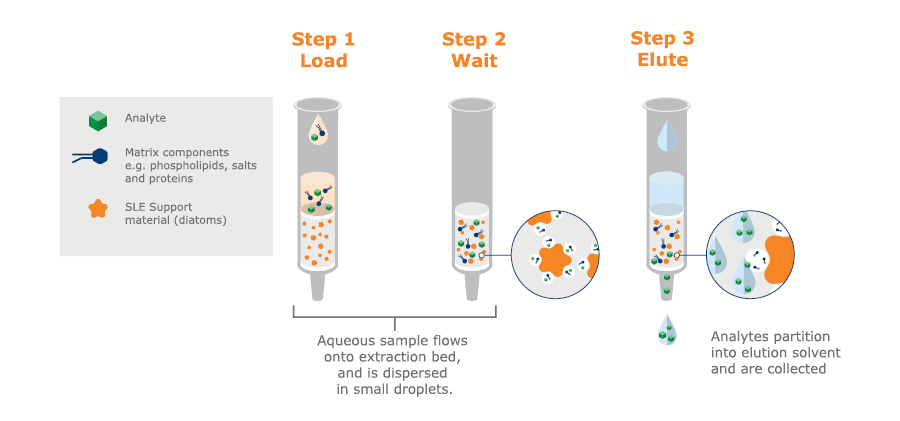
Fig. 7. Schematic SLE procedure available in 96-well plates or columns.
The outcome of SLE will always net an organic solvent, which one can direct inject or derivatize (for GC/MS analysis). If LC/MS analysis is used, then the solvent must be dried down and a compatible reconstitution solution must be employed, because LC-MS/MS normally uses reversed phase chromatography (RPC), which is incompatible with immiscible organic solvents.
Method development is required, but it is relatively simple.
Key variables are:
SPE is a very powerful targeted extraction technique that can combine sample clean-up and analyte concentration with high recoveries and low matrix effects (see figure 8). Micro-SPE combines these benefits with optimized small-scale formats to achieves high concentration analyte recoveries in small elution volumes.
However, method development in SPE is often more challenging than for the other techniques, and the overall greater complexity, both regarding solid phases and reagents, is reflected in higher costs per analysis.
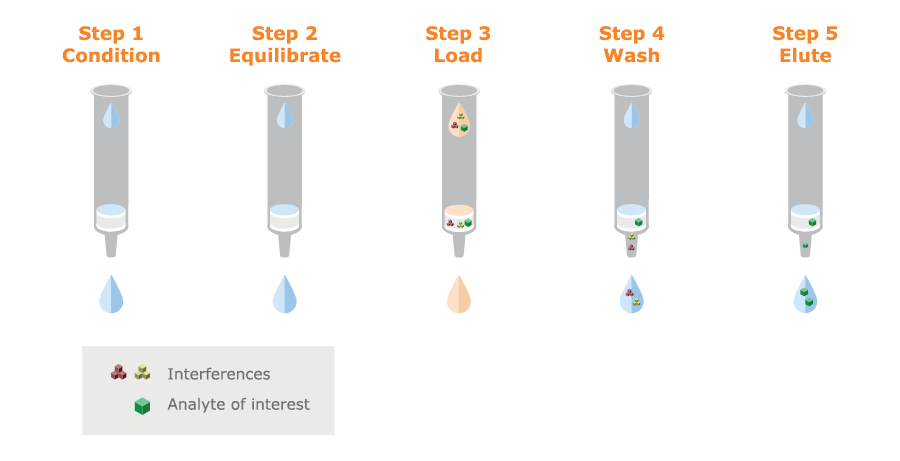
Fig. 8. Schematic SPE procedure. The silica or polymer particles can be packed in columns or a 96-well plate format.
SPE uses a solid phase to bind the analyte of interest under defined conditions. Matrix components are removed by one or several washing steps and the target is recovered by eluting under conditions that discourage binding.
The solid phase consists of modified silica or polymer particles that are packed in columns or 96-well plates. There is a wide choice of media offering many retention mechanisms and it is this wide choice that makes method development rather complex.
Silica-based SPE media characteristics
Non-polar modified silica: a wide range of hydrophobic bonded silicas are available, for example, C18, C8, C2, and PH.
Polar modified silica: these are not often used for biofluid extraction. However, non-bonded silica and amino- or diol-bonded silicas are available.
Cation exchange modified silica: cation exchangers can contain strong or weak acidic groups that give them different charge-carrying characteristics over a range of pH’s. A strong cation exchanger, such as one containing sulfonic acid groups, will be negatively charged over a wide pH range. A weak cation exchanger, such as one containing carboxylic acid groups, will be negatively charged at high pH values but will lose its charge gradually as pH values are reduced.
Anion exchange modified silica: In a similar fashion, a strong anion exchanger is one substituted with strong basic groups, such as quaternary amines, that are positively charged over a wide pH range. Weak anion exchangers typically contain amine groups that are positively charged at low pH but gradually lose their charge as pH is raised.
Mixed-mode modified silica: These types of media combine non-polar or hydrophobic characteristics with anion- or cation exchangers. The hydrophobic- and charge-based retention mechanisms result in solid phases that can retain most drugs and other small molecules. They have proved very useful for SPE of biological fluids since the combined retention mechanisms together with strong washes and selective elution solvent choices offer highly effective removal of matrix components, such as phospholipids, and good recovery of the specific analyte of interest.
The most common mixed-mode silica types are cation exchangers with either C8 or C18 non-polar groups. They are widely used in forensic toxicology applications to extract basic drugs from urine samples.
|
Sorbent type |
Retention mechanism |
Typical functional groups |
Used to extract |
|
Non-polar |
Hydrophobic |
C18, C8, C2, PH |
Neutrals, acids and/or bases, with some hydrophobic character |
|
Polar |
H-bonding, dipole-dipole or π-π |
-NH2, DIOL, |
Analytes with polar character (from non-polar solvents) |
|
Anion exchange |
Ion exchange |
Strong: -NR4+ Weak : -NH3+/-NH2 |
Acidic analytes with a negative charge |
|
Cation exchange |
Ion exchange |
Strong: -SO3- Weak : -COO-/-COOH |
Basic analytes with a positive charge |
|
Mixed-mode |
Combination of hydrophobic and ion exchange |
C8 + SO3-
C8 + -NR4+ |
Analytes with hydrophobic and ionic functional groups |
Table 2 Retention mechanisms of silica-based SPE media
Why use Polymeric SPE?
Polymeric SPE phases are often described as being more robust and are increasingly chosen in favor of silica-based media for biofluid extraction. This is because of a couple of drawbacks common to silica-based media:
pH Stability: silica-based media are not very stable above pH 8 and should only be used at higher pH’s (up to pH 10) for very short exposure times. In contrast, polymeric stationary phases, usually based on modified divinyl benzene (DVB) polymers, are stable over the pH range 1–14.
Conditioning: silica-based solid phases need to be ‘conditioned’ (usually by a wash with methanol) to ensure that the functional groups are active prior to use, and because of this, they can be sensitive to drying out. Polymeric SPE based on DVB are modified with polar monomers or functional groups to make them easily wettable without conditioning, and they are unaffected by drying out.
Polymer-based SPE media characteristics
Polymeric SPE particles consist of a backbone, typically a DVB modified with polar groups to make the polymer beads water wettable. These beads are then modified by substitution with ion exchange groups.
Mixed-mode polymeric SPE particles combine the characteristics of a non-polar (hydrophobic) polymer backbone (usually containing some polar/wettable groups) and an ionic functional group. In other words, they have mixed-mode binding characteristics, but this is achieved in a different way than with silica-based media.
A mixed-mode silica-based stationary phase tends to have lower hydrophobicity than the equivalent mixed-mode polymeric sorbent. Consequently, the non-polar retention mechanism provided by the silica-based C8 or C18 is generally less strong (less retentive) than that provided by a polymeric backbone. This can mean that cleaner extracts are possible from silica-based materials, because solvent washes can more effectively remove the weakly bound matrix interference components.
Polymeric SPE media
Typically, polymeric SPE stationary phases are available in 5 types:
Non-polar water wettable polymer: the primary retention mechanism is through non-polar interactions
Mixed-mode - strong cation exchange + non-polar water wettable polymer
Mixed-mode - weak cation exchange + non-polar water wettable polymer
Mixed-mode - strong anion exchange + non-polar water wettable polymer
Mixed-mode - weak anion exchange + non-polar water wettable polymer
Pore size considerations
The high surface area of SPE phases makes it possible to extract analytes using small amounts of media. The particles used in SPE, whether silica- or polymer- based, are highly porous, giving them a high surface area.
The size of the pores can influence extract cleanliness, particularly regarding proteins. With wider pores, more protein can penetrate inside of the particles, giving high binding capacity not only for the analyte but also for protein components that can interfere with the subsequent analysis.
In the experiment illustrated by the electrophoresis gel (see figure 9), an SPE sorbent with nominal 40 Å pore diameter is compared with one that has nominal 100 Å pores. Very little protein is extracted with the smaller pore size media.
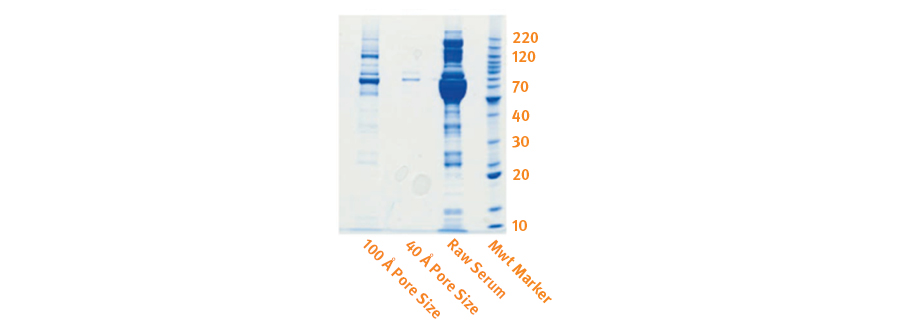
Fig. 9. Comparison of extracted protein using 40 Å and 100 Å pore diameter SPE media.
Biofluids are often diluted (usually 1:1 to 1:3, v:v) before the clean-up technique in order to:
Dilution in this way brings more consistency to samples that may vary considerably in viscosity — for example, urine samples can vary depending on how hydrated the patient is. Dilution will improve standardization, which is especially important when you are batch-processing or using 96-well plate formats.
All the sample preparation described, and the subsequent analytical techniques of LC-MS/MS or GC-MS, require liquid samples. Solids or semi solids must be pre-treated to get the analytes into the liquid state before clean-up. This is a very large area that we don’t cover here, but, for example, it can include:
Here we briefly summarize the special issues related to a few specific biofluids.
Urine
Urine contains urea, salts, creatinine, enzyme from hydrolysis, and sometimes other proteins associated with certain illnesses.
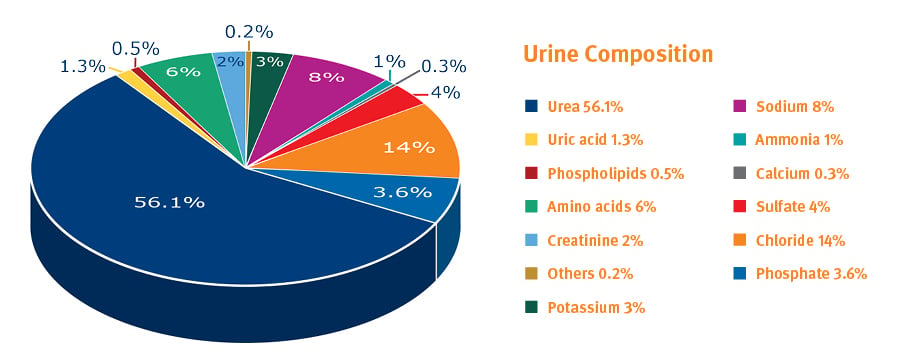
Fig. 10. Components in urine, excluding water (% values can vary considerably)1.
Hydrolysis to deconjugate drugs has been described in the section on Urine Hydrolysis. It improves detection limits for parent drugs (glucuronides, sulfates). Removal of post hydrolysis enzyme or other proteins is an important safeguard and, as described in the section on Eliminating Steps, a step in the workflow can be avoided using an in-well approach. Alternative hydrolysis approaches such as acid or base hydrolysis are also commonly used, and their impact on the sample prior to extraction (such as the effect of a changed pH) needs to be considered when developing sample preparation methods.
Whole blood/serum/plasma
Challenges with these types of samples can be divided into:
Lipid content can vary greatly depending on dietary differences and timing after meals.
Viscosity variations can also cause issues, for example related to vacuum manifolds, as mentioned here.
Some analytes, such as many drugs, are transported in blood bound to proteins and must be released prior to extraction using techniques such as SLE or SPE. Steps can include pH adjustment to denature the protein and addition of solvent to break the analyte – protein bond.
Oral fluid and associated collection devices
Things to consider here are if the sample is simply neat saliva or if it is collected with an oral fluid collection device that can introduce buffers and surfactants. These can impact on analyte recovery, or cause matrix effects in LC-MS/MS.
Others (hair/nails/meconium/tissue)
These solid or semi-solid samples introduce another level of matrix complexity that we will not address in detail here.
Collection of hair and nails is a non-invasive operation which is seeing growing popularity in drugs-of-abuse (DoA) analysis and workplace testing because sampling can be done in ’public’ (unlike with urine).
Other examples are meconium, which is used for monitoring DoA exposure in the womb, and liver tissue which is particularly useful in forensic postmortem cases where blood or urine may not be available.
As in any method development process, the first thing you should do is consider your lab’s purpose, your goals, and the measurable objectives that define achievement and good performance.
Scenarios can be very different in research, forensic and clinical environments. Ask the questions:
For example, incoming sample quality can be high and consistent, or low and variable.
Numbers of samples to be analyzed per day or week will steer the need for automation and the use of different formats and approaches.
Accuracy, precision and sensitivity are all important parameters for analytical work, but they might be prioritized differently depending upon the application. Normally you can’t have it all, and there will be a trade-off between simplicity, speed and cost.
For many sample preparation methods, there is a basic choice between columns (or test tubes) and 96-well plates, the former allowing larger sample volumes, the latter offering greater opportunities for improved throughput and automation, with lower elution volumes and, usually, smaller sample volumes.
PPT
In devices with a bottom filter, flow can be induced by positive pressure or vacuum to remove the precipitate. Precipitate can also be spun down by centrifugation, although the workflow for multiple samples is difficult to automate.
LLE and SLE
LLE is a very hands-on procedure, with equipment varying from large, traditional separation funnels, through disposable capped tubes and micropipettes, to 96-well plates. In addition, LLE can often be replaced by SLE, which has greater potential for automation.
SPE
SPE is available in columns, standard 96-well plates- or micro-SPE plate formats. Your choice of column or 96-well plate format depends on the typical number of samples you want to process per batch, the volume of sample to be extracted, and your overall lab throughput. Choosing a micro-SPE plate format may be governed by you having very small sample volumes, or by your need to achieve small and concentrated elution volumes – thereby gaining sensitivity in the subsequent analysis.
To induce flow, the simplest manual method is to connect an SPE column to a syringe barrel and, by depressing the barrel, force the liquid (solvent or sample) through the SPE column. This is cheap and effective, but not convenient for processing multiple samples.
Vacuum manifolds are often used to process samples in parallel (for example 10-20 columns or 96 samples in a plate format). Liquids can be manually added by the operator, who also controls the vacuum, although automated systems are also available (see here).

Fig. 11. Vacuum manifold for manual processing of SPE columns
Vacuum manifolds are low cost and easy to use but there are challenges, such as plugged wells/columns due to particulate materials in the samples that can result in the faster-flowing wells/columns running dry and creating free-flow paths for the vacuum to pull through.
A similar challenge with vacuum-induced flow is that as less-viscous samples vacate their wells quickly, the pressure drop over all the wells decreases, and this can leave more-viscous samples still on the plate, resulting in variable analyte recoveries.
These problems can be avoided by using positive pressure systems to process multiple samples in parallel. Common formats are 96, 48 or 24 columns, depending on the column size, or 96-well plate devices. Again, liquids can be manually added by the operator, who also controls the pressure. Alternatively, fully automated systems operating with positive pressure are available.

Fig. 12. Positive pressure manifold for 96-well plate processing
Every additional step:
Lean operation principles are applicable in sample preparation, just as they are when considering total lab functions, or automobile manufacturing!
Looking at table 1 again, we see that many sample preparation techniques can be steered to allow direct injection into the analytical system, without extra steps — the exceptions being LLE and SLE.
Also, evaporation and reconstitution can be avoided if sample prep can be achieved with SPE.
And, of course, if applicable, D&S and filtration require fewer steps than other sample preparation techniques, but they neither eliminate matrix effects nor ‘extract’ the analyte.
A smart example of step elimination is ‘in-well hydrolysis’ during DME or SPE, which eliminates a sample transfer step. This is achievable with the help of special hydrophobic frits that hold up the sample during hydrolysis but allow it to pass through when pressure is applied after hydrolysis. Hydrolysis efficiency is not affected by performing the hydrolysis in this way.
In SPE, the use of a water-wettable polymer-based media eliminates a conditioning step that is required with silica-based media (compare figure 13 with figure 8).
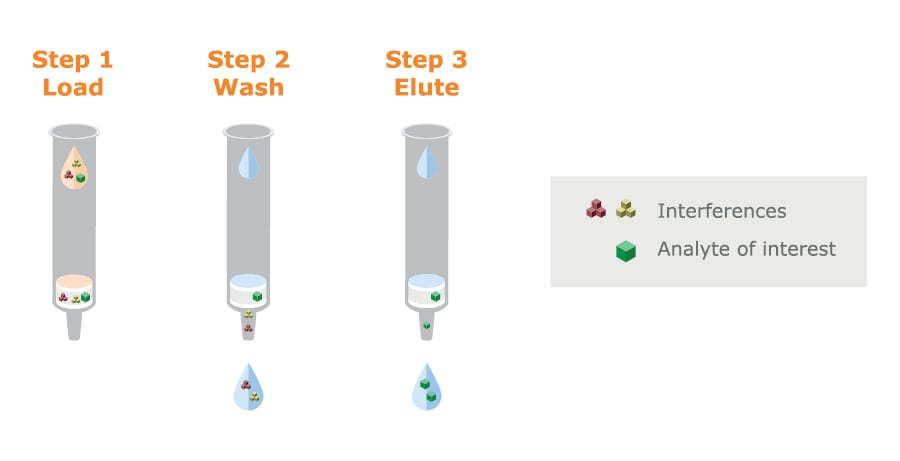
Fig. 13. Using a wettable polymer-based SPE adsorbent eliminates the conditioning and equilibration steps that are required with silica media.
Automation will reduce the risk for human errors and allow a higher throughput in your lab, but it will require investment in equipment. However, running costs per analysis should be less after automation and highly trained staff can be used for more demanding tasks than repeated manual operations.
It is important to consider the whole workflow so that automation of sample preparation or even a single step within sample preparation, doesn’t simply lead to a bottle neck at some other point.
There are 2 major choices in automation of sample prep:
Both approaches have their advantages.
For further information about automated sample preparation look here.
Vacuum versus positive pressure processing in automated sample preparation
Flow-through sample preparation techniques such as SPE, PPT, SLE and filtration are often automated. A high throughput lab will prefer to do parallel processing automatically.
The choice between a positive pressure or vacuum SPE system is particularly important for automated processing. With 96-well plates and for micro-SPE a blocked well or a well with remaining sample may not be detected. Vacuum manifolds for columns can have the option of individual column control, so that you can ‘switch off’ the vacuum for columns that have run dry, BUT this requires constant monitoring and is not conducive to high throughput, and can’t be easily automated.
Positive-pressure based automation systems eliminate these issues.
Sample prep helps you clean-up biological samples before analysis, but it doesn’t automatically improve sensitivity. Sometimes, only small amounts of sample are available. High-sensitivity analysis by LC-MS/MS demands the injection of small volumes of sample extract. High concentrations of the analyte that is sometimes in short supply are needed to achieve low detection limits. So, the lower the extract volume the more concentrated the extract, and the lower the detection limit that can be achieved.
Table 3 indicates sample prep techniques that allow convenient direct injection into an LC-MS/MS system, the ones that dilute the analyte and need to be combined with evaporation and reconstitution, and the ones that offer potential concentration. The table shows where you might lose or gain sensitivity.
Matrix scavenging methods and LLE/SLE usually dilute the analyte and, if additional sensitivity is needed, you must evaporate and reconstitute to achieve high concentrations. In contrast, SPE inherently concentrates the analyte.
|
Technique |
Inherent concentration? |
Direct injection possible? |
Evaporation + reconstitution? |
|
D&S |
No, sample is diluted |
Yes |
N/A |
|
Filtration |
No |
Yes |
N/A |
|
PPT |
No, sample is diluted |
Yes, but dilution with mobile phase may be necessary for compatibility with LC-MS/MS conditions |
Yes, for low detection limits |
|
PLD |
No, sample is diluted |
Yes, but dilution with mobile phase may be necessary for compatibility with LC-MS/MS conditions |
Yes, for low detection limits |
|
DME |
No, sample is diluted |
Yes, but dilution with mobile phase may be necessary for compatibility with LC-MS/MS conditions |
Yes, for low detection limits |
|
LLE & SLE |
No, sample is usually diluted |
No, extraction solvent is incompatible with reversed phase LC-MS/MS |
Yes. Essential |
|
SPE |
Usually |
Yes, but dilution with mobile phase may be necessary for compatibility with LC-MS/MS conditions |
Yes, for low detection limits |
|
Micro-SPE |
Yes |
Yes, but dilution with mobile phase may be necessary for compatibility with LC-MS/MS conditions |
No |
Table 3. Sample prep characteristics that impact detection limits and workflow streamlining opportunities.
Although evaporation followed by reconstitution in a smaller volume of solvent is a standard approach, it has several challenges.
The evaporation step can take time and increases the complexity of sample preparation. Some analytes are also prone to losses during evaporation, which can reduce sensitivity, and make it difficult to achieve the required detection limits. This is particularly problematic for:
Micro-SPE devices are essentially miniature versions of the traditional 96-well SPE plate that contain tiny amounts of sorbent. However, this is not enough to assure high concentrations of eluted analyte with small samples. This is achieved by eliminating dead volumes and using relatively long and narrow columns of SPE stationary phase, rather than short wide ones.
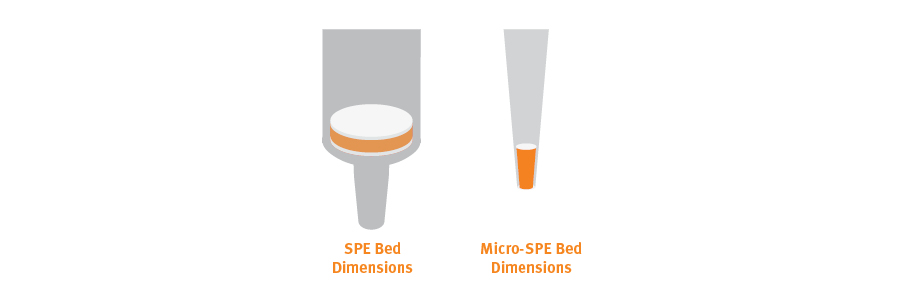
Fig. 14. Micro-SPE bed dimensions are taller and narrower than standard SPE
Micro-SPE can help avoid evaporation and reconstitution, saving time and offering high recovery of analytes in small elution volumes — ready for high sensitivity LC-MS/MS.
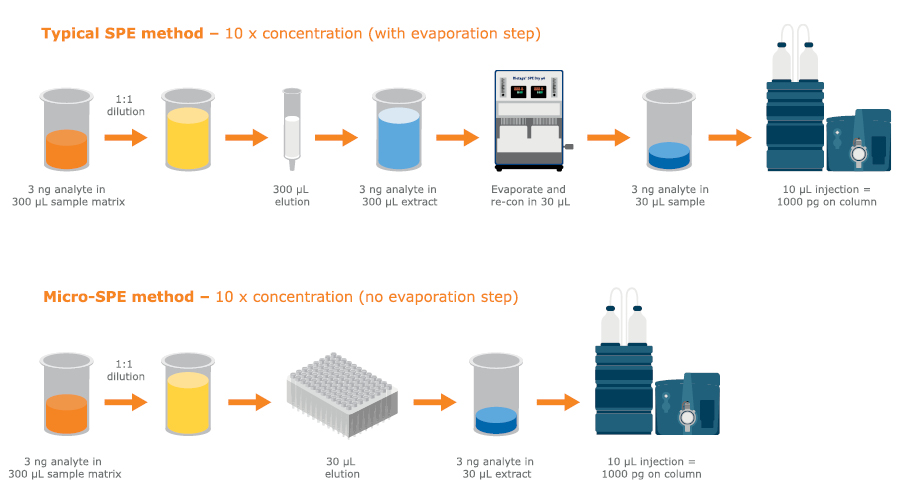
Fig. 15. Low elution volumes in micro-SPE can help avoid evaporation steps
Sample preparation offers significant benefits if you are using sophisticated analytical techniques such as LC-MS/MS and GC-MS but finding the right solution to meet your needs can be complex. This brief overview hopefully illustrates the range of challenges and the choices available. You should speak with your peers and talk to manufacturers of sample preparation consumables and systems, who often have a deep understanding of the issues. Arrange to see various options in action, especially if you are thinking of high throughput situations and automation. Understand how equipment will operate in practice and note the difficulties as well as the benefits. In the end, your lab and your colleagues will benefit from a good choice of this ‘necessary inconvenience’.
If you found this useful, why not download for future reference?
Biotage is a Global Impact Tech Company committed to solving society’s problems. We offer workflow solutions and products to customers in Drug Discovery and Development, Analytical Testing and Water and Environmental Testing.