Extraction of antiepileptic drugs from serum and urine using ISOLUTE® SLE+ prior to LC-MS/MS analysis
This application note describes the extraction of neutral and zwitterionic antiepileptic drugs fortified in serum and urine using ISOLUTE® SLE+ in a 96-well plate format.
Figure 1. Structures of Carbamazepine Epoxide, Rufinamide and Gabapentine
Introduction
Antiepileptic drugs (AED) are prescribed to suppress seizures in epilepsy patients. A variety of different types of AEDs have been synthesized to pharmacologically address different types of epilepsy. The ability to therapeutically monitor these drugs in patients is necessary for maintaining optimal medical care and managing any adverse effects of the drug. A fast and clean extraction method is needed that works in a variety of biological matrices and affords a high throughput workflow. Here ISOLUTE SLE+ is demonstrated as an effective way to extract AEDs from serum and urine with good efficiency. The method was developed using a 96-well plate format to facilitate a high throughput workflow model.
ISOLUTE SLE+ Supported Liquid Extraction plates and cartridges offer an efficient alternative to traditional liquid-liquid extraction (LLE) for bioanalytical sample preparation, providing high analyte recoveries, no emulsion formation, and significantly reduced sample preparation time.
Analytes
Tiagabine, Carbamazepine 10,11 epoxide, Oxcarbazepine, Gabapentin, Vigabatrin, Rufinamide and Felbamate
Sample preparation for neutral antiepileptic drugs in serum and urine
Format: ISOLUTE® SLE+ 400 μL 400 Supported Liquid Extraction plate, part number 820-0400-PO1
Sample pre-treatment: Pipette serum/urine (blank, calibrator or patient (100 µL)) into a container. Add ammonium acetate (5mM, pH 2.9, 250 µL). Add up to 50 µL of internal standard. Mix.
Sample processing: Load up to 400 µL of pre-treated serum/urine sample onto the ISOLUTE SLE+ 96-well plate. Apply a short pulse of positive pressure or vacuum and allow samples to sit for 5 minutes.
Analyte elution: Elute analytes with methyl tert-butyl ether containing 1% (v/v) trifluoroacetic acid (conc) solution (2 x 700 µL). Allow sample to flow through by gravity and collect eluent. Apply positive pressure or vacuum as needed to facilitate a constant flow of approximately 1 mL/min (10–12 drops).
Sample preparation for neutral antiepileptic and zwitterionic drugs in serum and urine
Format: ISOLUTE® SLE+ 400 μL 400 Supported Liquid Extraction plate, part number 820-0400-PO1
Sample pre-treatment: Pipette serum/urine (blank, calibrator and patient (100 µL)) into a container. Add 50% aqueous formic acid (100 µL). Add up to 100 µL of internal standard.
Sample Processing: Load up to 300 µL of pre-treated serum sample onto the ISOLUTE SLE+ 96-well plate. Apply a short pulse of positive pressure or vacuum and allow samples to sit for 5 minutes.
Analyte elution: Elute analytes with methyl tert-butyl ether containing 1% (v/v) trifluoroacetic acid (conc) solution (2 x 700 µL). Allow sample to flow through by gravity and collect eluent. Apply positive pressure or vacuum as needed to facilitate a constant flow of approximately 1 mL/min (10–12 drops).
Post-extraction all: Evaporate to dryness (45 °C for 15 mins) and reconstitute sample in mobile phase.
HPLC conditions
Instrument: Agilent 1200 Liquid Handling System (Agilent Technologies, Berkshire, UK)
Column: Phenomenex Gemini C18, 150 mm x 4.6 mm (5 µm)
Mobile phase: Solvent A: 5mM Ammonium Formate with 0.01% (v/v) Formic Acid Solvent
B: Methanol: Acetonitrile (50:50m v/v)
Gradient:
|
Step |
Time (min) |
Flow Rate (mL/min) |
%A |
%B |
|
1 |
0.0 |
1000 |
70 |
30 |
|
2 |
0.50 |
1000 |
70 |
30 |
|
3 |
3.0 |
1000 |
30 |
70 |
|
4 |
4.0 |
1000 |
30 |
70 |
|
5 |
4.5 |
1000 |
70 |
30 |
|
6 |
7.5 |
1000 |
70 |
30 |
Mass spectrometry conditions
Instrument: Applied Biosystems/MDS Sciex 4000 Q-Trap triple quadrupole mass spectrometer (Applied Biosystems, Foster City, CA.) equipped with a Turbo Ionspray® interface for mass analysis.
Ion Source Temperature: 700 °C
|
Analyte |
MRM Transition |
Declustering Potential (DP) |
Collision Energy (CE) |
Cell Exit Potential (CXP) |
|
Gabapentine |
172>154 |
40 |
25 |
16 |
|
Felbamate |
178>117 |
40 |
25 |
16 |
|
Rufinamide |
239>127 |
40 |
25 |
16 |
|
Oxcarbazepine |
253>208 |
40 |
25 |
16 |
|
Tiagabine |
376>247 |
40 |
25 |
16 |
|
Vigabatrine |
130>70.9 |
40 |
25 |
16 |
|
Carbamazepine Epoxide |
253>180 |
40 |
30 |
16 |
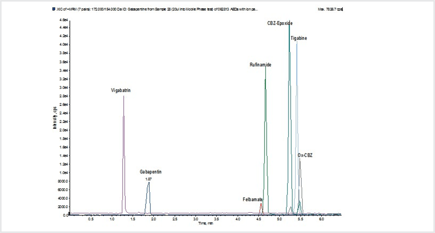
Figure 2. Extracted ion chromatogram of antiepileptic drugs
Results and discussion
Antiepileptic drug (AED) stock standards were prepared in methanol: acetone (80:20, v/v) at 100 µg/mL concentration. The parameters for LC-MS/MS analysis were identified and optimized using the stock solutions (Figure 2). The stock solutions were fortified into blank urine and serum matrix at final spiking concentrations of 20 ng/mL. Two individual pre-treatment strategies were developed to optimize extraction recoveries for the AEDs as a function of their polarity. The elution strategy utilizes methyl tert-butyl ether mixed with 1% concentrated trifluoroacetic acid (TFA) (v/v) to yield optimal recoveries using either of the pre-treatment strategies. The addition of TFA to the extraction solvent seemed to increase overall recoveries by 5–10%.
The first pre-treatment strategy calls for 5 mM ammonium acetate to be added to the fortified matrix at a 1:2.5 (v/v) ratio. Recoveries for the AEDs using this pre-treatment strategy were good for all of the neutral AEDs in urine and serum and lower for the zwitterionic AEDs, particularly in the serum matrix (Figure 3). The lower recovery for the gabapentin and vigabatrin was attributed to their lower log P values (<1).
The second pre-treatment strategy calls for the addition of a 50% aqueous formic acid buffer to the fortified matrix at a 1:1 (v/v) ratio. This particular strategy increases the recovery for the gabapentin significantly. (Figure 4) The vigabatrin does not benefit from formic acid pre-treatment. The neutral AED recoveries were good using this strategy. It should be noted that a decrease in the extracted ion peak intensity for the carbamazepine epoxide was lower in the presence of the 50% formic acid. This was attributed to the epoxide ring opening to yield the hydroxylated analogue.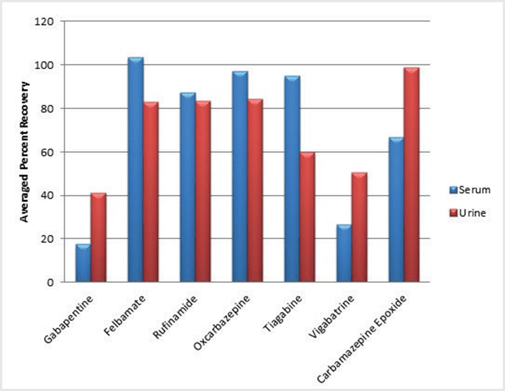
Figure 3. Plot of average recoveries for neutral antiepileptic drugs (n=7) and zwitterionic antiepileptic drugs (n=4) fortified into serum and urine at 20 ng/mL and pre-treated with 5 mM ammonium acetate.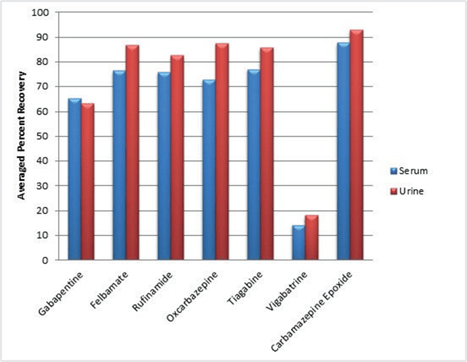
Figure 4. Plot of average recoveries for neutral and zwitterionic antiepileptic drugs (n=7) fortified into serum and urine at 20 ng/mL and pre-treated with 50% aqueous formic acid.
Ordering information
|
Part Number |
Description |
Quantity |
|
820-0400-P01 |
ISOLUTE® SLE+ 400 μL Supported Liquid Extraction Plate |
1 |
|
PPM-96 |
Biotage® PRESSURE+ 96 Positive Pressure Manifold |
1 |
|
SD-9600-DHS-EU |
Biotage® SPE Dry Sample Concentrator System 220/240 V |
1 |
|
SD-9600-DHS-NA |
Biotage® SPE Dry Sample Concentrator System 100/120 V |
1 |
Literature number: AN805

