Extraction of vitamins B1 and B6 from whole blood using ISOLUTE® PLD+
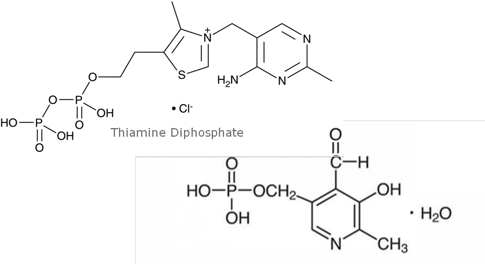
Figure 1. Structures of analytes.
Introduction
Vitamin B1 (Thiamine Diphosphate, TDP) and B6 (Pyridoxal-5- Phosphate, PLP) in whole blood is measured clinically to help measure various metabolic functions. TDP and PLP are highly water soluble (logP= -5.9, -2.2 respectively) and are extremely light-sensitive. Traditionally, TDP and PLP are measured through fluorescence detection, requiring the use of hazardous reagents for derivatization. This additional derivatization step is also time consuming. This application note describes a simple, ISOLUTE® PLD+ protocol for the extraction of vitamin B1 and B6 (TDP/PLP) from whole blood and LC-MS/MS analysis.
High, reproducible recoveries and low protein and phospholipid coextraction were demonstrated. ISOLUTE PLD+ Protein and Phospholipid Removal plates and cartridges offer a fast and extremely simple sample preparation approach. Proteins are efficiently removed from the sample using an in-plate, ‘solvent first’, protein crash and filter procedure, which is easily automated.
Analytes
Vitamin B1(Thiamine Diphosphate, TDP) and Vitamin B6 (Pyridoxal-5-Phosphate, PLP).
Sample preparation procedure
Format
ISOLUTE® PLD+ Protein and Phospholipid Removal Plate
Processing
Samples are processed manually using the Biotage® PRESSURE+ 96 positive pressure manifold. Each step described below was processed at 6 to 9 psi using the adjustable flow setting. Drying was performed using the TurboVap® 96 Dual 50 L/min flow at 4ooC (gas) and 60 oC (plate) with a plate height of 55 mm.
Extraction procedure
Solvent application
Apply 330 µL of precipitation solvent (0.1 M HCl + 1.2 M perchloric acid) to each well of the ISOLUTE® PLD+ plate.
Sample application
Add 200 µL of whole blood to each well via handheld pipette and mix thoroughly via 5 repeat aspirate/dispense steps.
Analyte elution
Apply positive pressure (10 psi) for approximately 5 minutes. For highly particulate laden or viscous samples, increased pressure may be required. Collect eluent in a square 2 mL 96-well plate (p/n 120-5203).
Post-extraction (optional)
While samples can be analyzed directly after extraction, HCl is known to be corrosive to the MS source, so samples should be dried and reconstituted in 0.1 % formic acid. Dry the extract in a stream of air or nitrogen using a TurboVap® 96 Dual 96 position manifolds in single or dual mode (for 2 plates) with 50 L/min flow at 40 °C (gas) and 60°C (plate) with a plate height of 55 mm. If necessary, transfer samples via pipette to autosampler vials.
Reconstitution
Reconstitute in 200 µL 0.1% formic acid and mix well.
Analytical conditions
UHPLC conditions instrument:
Shimadzu Nexera X2
Column
Phenomenex Gemini 5 µm C18 (100 x 3 mm id)
Mobile phase
A: 10 mM Ammonium bicarbonate pH 8.8 (aq)
B: Methanol
Flow Rate: 0.6 mL/min
|
Time (min) |
%A |
%B |
|---|---|---|
|
0.01 |
100 |
0 |
|
1.5 |
100 |
0 |
|
5 |
40 |
60 |
|
6.5 |
40 |
60 |
|
6.51 |
100 |
0 |
|
9 |
100 |
0 |
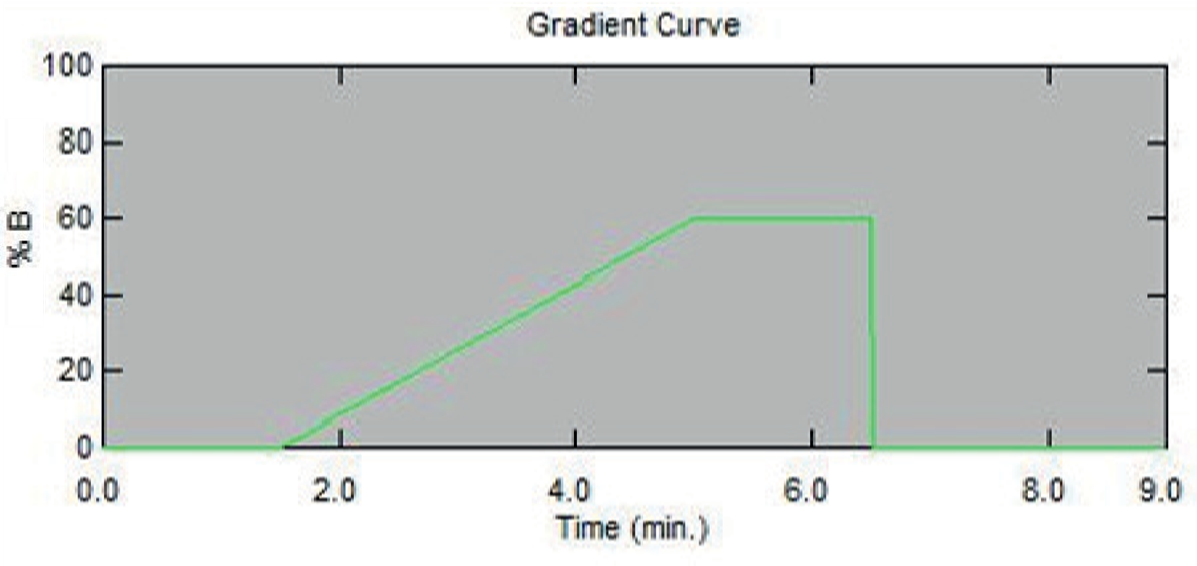
Figure 2. HPLC Gradient Conditions.
MS/MS conditions
Instrument
Sciex 5500 MSD
Ion source temperature
425°C
|
ID |
Q1 |
Q3 |
DP (volts) |
CE (volts) |
CXP (volts) |
|---|---|---|---|---|---|
|
PLP1 |
248.0 |
150.1 |
40 |
25 |
6 |
|
PLP2 |
248.0 |
122.1 |
80 |
30 |
6 |
|
PLP3 |
248.0 |
94.1 |
60 |
40 |
7 |
|
TDP1 |
425.0 |
304.0 |
80 |
20 |
8 |
|
TDP2 |
425.0 |
81.0 |
60 |
25 |
7 |
|
TDP3 |
425.0 |
122.0 |
80 |
20 |
7 |
Results
The negative log P value for TDP (-5.9) and PLP (-2.2) allows for the aqueous acidic crash solvent to double as the extraction solvent. Recoveries based on endogenous levels of TDP/PLP are presented in Table 3.
Chromatography
Good separation was observed between the TDP/PLP. Figure 3 represents spiked whole blood extraction of TDP/PLP at 75 ng/mL.

Figure 3. Spiked whole blood extraction of TDP/PLP at 75 ng/mL.
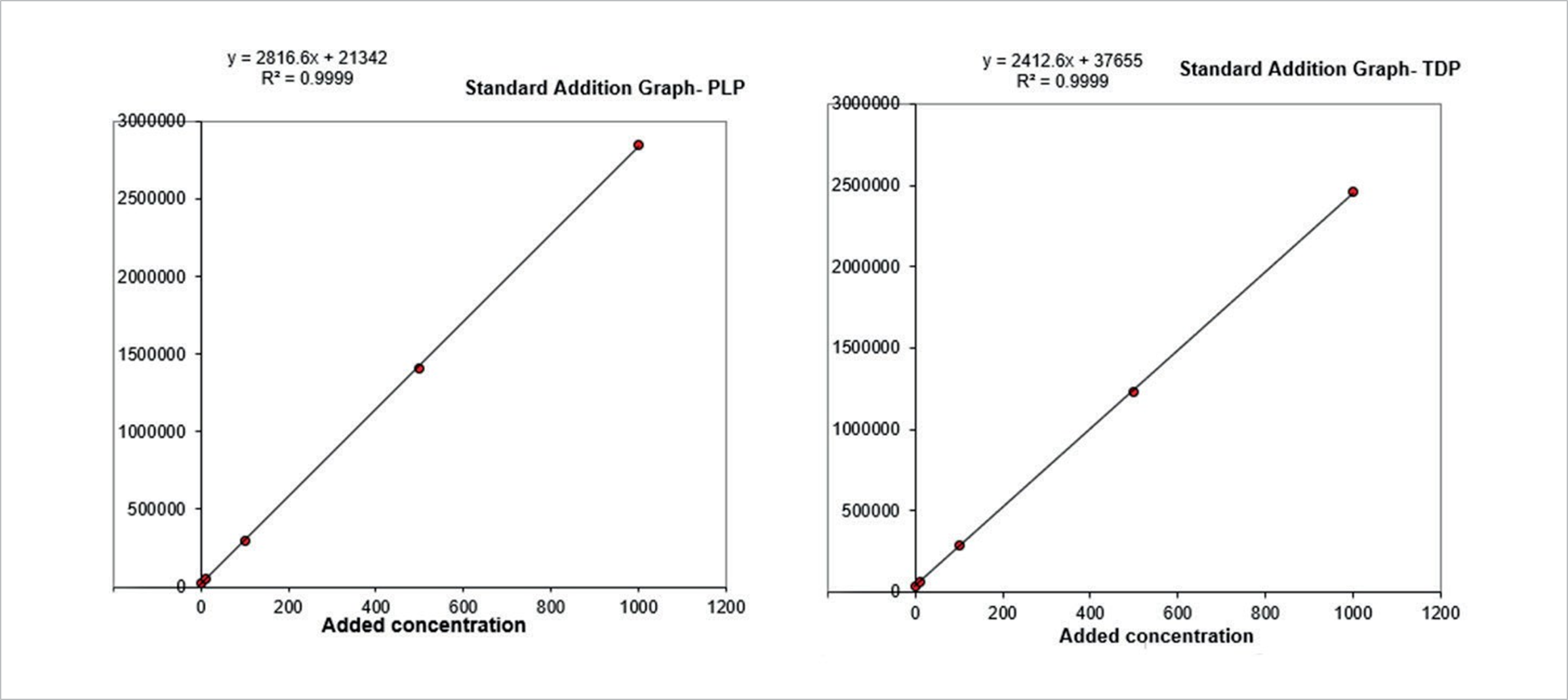
Figure 4. Standard addition calibration curves for PLP and TDP.
Calibration curves
Stripped whole blood was not available at the time of the study, so standard addition calibration was performed in a range of 10–1000 ng/mL. Good linearity was observed (r2>0.99) over the calibration range. Figure 4 shows representative standard addition calibration curves.
Analyte recovery
Whole blood samples prepared by precipitation alone (no ISOLUTE® PLD+ sample prep) were compared to samples prepared with ISOLUTE PLD+ plates as described previously. The average of 5 injections were recorded and percent recovery and RSD values were obtained. Percent recovery is based upon the calculated recovery of the TDP/PLP by precipitation alone, highlighting any analyte loss due to the use ISOLUTE PLD+ sample preparation plate. Table 3 summarizes the recovery data.
|
Analyte |
Precipitation alone (ng/mL) |
PLD+ (ng/mL) |
Recovery % |
% RSD (n=5) |
|
PLP |
43.7 |
36.4 |
81.9 |
1.2 |
|
TDP |
83.5 |
78.6 |
93.9 |
1.3 |
Phospholipid and protein removal
The efficacy of ISOLUTE PLD+ Phospholipid and Protein Removal Plate was investigated via LC-MS/MS analysis of residual phospholipids and gel electrophoresis analysis for proteins. Figure 5 shows no detectable protein in electrophoresis data for sample treated with ISOLUTE PLD+. A significant reduction (>99%) of phospholipids is observed in the LC-MS/MS analysis of the common 184 product ion (Figure 6) when compared to whole blood precipitated alone.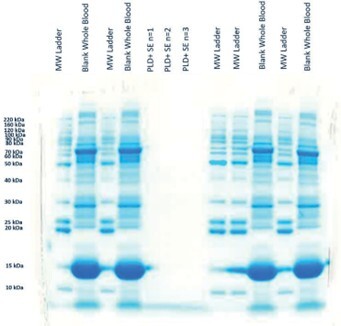
Figure 5. Gel Electrophoresis on whole blood precipitated with aqueous acidic crash solvent and using ISOLUTE® PLD+.
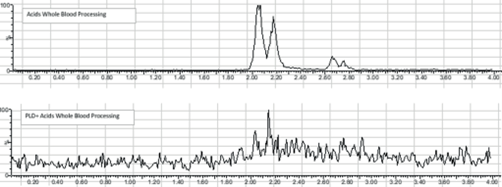
Figure 6. LC-MS/MS on whole blood precipitated with 100% aqueous acidic crash solvent and ISOLUTE® PLD+. The 184–184 transition was monitored in precipitated whole blood (top) and whole blood precipitated on ISOLUTE® PLD+ (Bottom).
Conclusion
Whole blood is precipitated and vitamin B1(Thiamine Diphosphate, TDP) and B6 (Pyridoxal-5-Phosphate, PLP) are extracted in the same acidic solution using the ISOLUTE® PLD+ Protein and Phospholipid Removal Plate. No protein was detected in ISOLUTE PLD+ treated samples via gel electrophoresis and phospholipids were reduced >99%. When compared to samples precipitated alone, the PLD+ plate has good recovery of TDP (93.9%) and PLP (81.9%) with %RSD less than 2.
Additional notes
- Mobile phase A (10 mM ammonium bicarbonate, pH 8.8) preparation: add 0.8024 g ammonium bicarbonate to 1 L of HPLC grade water. Adjust pH to 8.8 with ~2.3 mL of 5% ammonium hydroxide.
- This application note was developed using the specified matrix. Further optimisation may be required for other similar matrices (for example, to compensate for regional dietary variation).
- Internal standards were unavailable (COVID, supply chain) at the time of this study but have been requested and ordered.
- The work presented was performed in a 96-well plate format, if desired, ISOLUTE® PLD+ Protein and Phospholipid Removal cartridges are available (p/n 918-0005-AG).
Ordering information
|
Part Number |
Description |
Quantity |
|
918-0050-P01 |
ISOLUTE® PLD+ Protein and Phospholipid Removal Plate |
1 |
|
918-0005-AG |
ISOLUTE® PLD+ Cartridges 50 mg/1 mL (tables) |
100 |
|
121-5203 |
Collection Plate, 2 mL, Square |
50 |
|
PPM-96 |
Biotage® PRESSURE+ 96 Positive Pressure Manifold |
1 |
|
418000 |
TurboVap® 96 Dual |
1 |
Literature number: AN985

