Extraction of acrylamide from coffee using ISOLUTE® SLE+ prior to LC-MS/MS analysis
By Biotage
Introduction
The method described in this application note achieves high recoveries of acrylamide in coffee. The method is sensitive enough to measure levels as low as 1 ng/mL in coffee (solution), 25 ppb (25 ng/g) in ground coffee (solid) or 125 ppb (125 ng/g) in instant coffee (solid, traditional or decaffeinated) and gives good selectivity from what is a challenging matrix. ISOLUTE SLE+ products provide clean, rapid, robust and efficient extraction solutions for a wide range of analytes.
Analytes
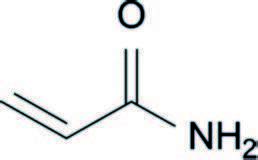
Figure 1. Acrylamide
Sample preparation procedure
Sample extraction experiments were conducted with non-smoker donor urine (pH 6.30) for the recovery of TSNAs. Synthetic urine (UriSub, pH 7.4) was used for calibration solutions to avoid interference from human urine which, may contribute to the TSNA signal.
Format
ISOLUTE® SLE+ 1 mL Columns (Tabless), part number 820-0140-CG
Sample pre-treatment
Coffee was prepared in the same way that it would normally be consumed. In the case of ground coffee, 60 g of ground coffee was percolated with 1500 mL of boiling water. For instant coffee 2 g of instant coffee powder was dissolved in 250 mL of boiling water. This resulted in solutions containing coffee ‘solid’ concentrations of 40 mg/mL for ground coffee and 8 mg/mL for instant coffee. Once prepared the coffee was left to reach room temperature.
Calibration line preparation:
A 128 ng/mL acrylamide coffee over-spiked solution was prepared by diluting 25.6 μL of a 10 μg/mL aqueous acrylamide solution to 2 mL with control coffee.
This was then serially diluted seven times by transferring 0.8 mL, diluting with 0.8 mL of control coffee, mixing, and then transferring 0.8 mL of this mixture; repeating the procedure until a solution with an over-spiked level of 1 ng/mL had been reached.
0.625 mL aliquots were transferred to wells containing 10 μL of a 4 μg/mL 13C3 acrylamide solution in water and 12.75 μL of a saturated solution of ammonium hydroxide in water.
Supported liquid extraction
Sample work-up
Samples (0.625 mL) were transferred to tubes containing 10 μL of a 4 μg/mL 13C3 acrylamide solution in water and 12.75 μL of a saturated solution of ammonium hydroxide in water. The tube was briefly shaken and then 0.5 mL of the mixture transferred to a 1 mL capacity ISOLUTE® SLE+ column.
Sample loading
Load pre-treated sample (0.5 mL) onto each well. Apply a pulse of vacuum (VacMaster-10 or 20 Sample Processing Manifold, 121-1016 or 121-2016) or positive pressure (Pressure+ Positive Pressure Manifold, PPM-48) to initiate flow. Allow the sample to absorb for
5 minutes.
Analyte elution
Elute with ethyl acetate: tetrahydrofuran, (1 : 1, v/v, 2x 2.5 mL) and allow to flow under gravity into a tube already containing 2 μL ethylene glycol. Apply vacuum or positive pressure to elute any remaining extraction solvent.
Post elution
Dry the volatile constituents of the eluate in a stream of air or nitrogen using a TurboVap LV (415000). Reconstitute in water (200 μL).
HPLC conditions
Instruments
Waters Acquity
Column
Phenomenex Hydro, 4 μm 50 x 2 mm C18 column with a C18 guard cartridge and on-line filter
Mobile Phase
A: 0.1% formic acid in water
B: 0.1% formic acid in methanol
Flow rate
0.3 mL min-1
Injection
10 μL
Gradient
Initial 100 % A, hold till 0.6 min
linear ramp to 100 % B over 0.25 min (0.85 min), hold 1.65 min (2.5 min)
linear ramp to 100 % A in 0.01 min (2.51 min), hold 2.49 min (5 min)
Column temperature
40 °C
Sample temperature
20 °C
Table 1. Typical retention times for acrylamide using the LC-MS/MS method described
|
Retention time (min) |
|
|
Acrylamide |
1.02 |
|
Acrylamide13C3 |
1.02 |
MS conditions
Ions were selected in order to achieve maximum sensitivity using multiple reaction monitoring.
Instrument
Waters Quattro Premier
Ionization mode
ES+
Desolvation temp.
450 °C
Source temp
120 °C
Table 2: Positive ion mode - MRM parameters
|
MRM transition |
RT |
Compound ID |
Cone, V |
CE, V |
|
71.9 - 55.2 |
1.0 |
Acrylamide |
23 |
8 |
|
74.9 - 58.2 |
1.0 |
Acrylamide 13C3 |
24 |
9 |
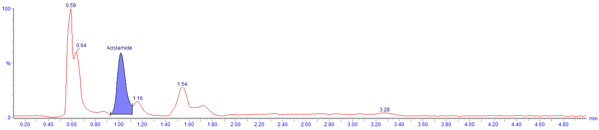
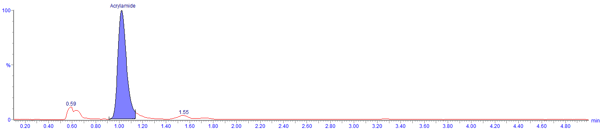
Figure 1. Extracted ion chromatograms in positive ion mode using
ISOLUTE® SLE+ procedure (sample: 500 μL ground coffee, not spiked
(process derived levels only) and over-spiked with 128 ng/mL acrylamide)
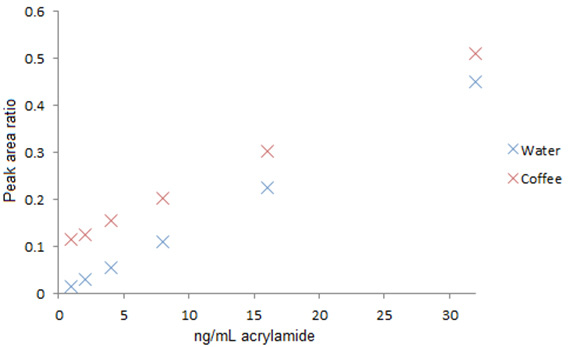
 Figure 2. Typical calibration curve for acrylamide in ground coffee,
Figure 2. Typical calibration curve for acrylamide in ground coffee,
expressed on a linear scale.
Table 3. Performance and recovery data for acrylamide
| Matrix | Recovery % | % RSD (n=6) |
| Fresh roast coffee | 81 | 8.2 |
| Instant coffee | 82 | 5.5 |
| Instant decaffeinated coffee | 73 | 3.7 |
Recovery and RSD calculations based on extractions of blank
matrix spiked at 64 ng/mL without using an internal standard.
The blank acrylamide response was subtracted from both extracted and fortified quantities prior to calculating both recovery and RSD.
Table 4. Analyte performance from ground coffee
| Analyte | r2 |
| Acrylamide | 0.998 |
r2 calculations were based on line including a ‘zero’ standard,
over-spiked standards between 1 to 128 ng/mL and applying
a weighting factor of 1/x.
Additional notes
The addition of the ammonia solution results in the coffee changing from a mid-brown to a darker brown appearance. Although the sample added to the ISOLUTE SLE+ column is darker as a result of the basification, the final extract appears visibly cleaner than with
untreated coffee. The image below shows 0.5 mL untreated ground coffee (left), coffee combined with 2% concentrated ammonia solution (middle) and the SLE extract diluted to an identical volume with water (right).

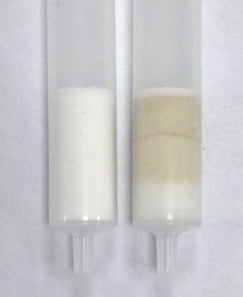
The majority of the coffee dyes are removed by being trapped on the SLE material. The image below shows an unused 1 mL SLE column (left) compared to a column that has undergone a full extraction including the removal of acrylamide (right).
A calibration line extracted in water gave similar properties to that extracted in coffee but without the intercept due to the lack of any process derived acrylamide. Preparing a calibration line in this solvent could be applied for ultra low level acrylamide determinations.
Ethylene Glycol was added in a small quantity prior to the extraction step to avoid the evaporated sample drying completely. Without this additive being present the majority of the acrylamide would be lost at this stage.
A 100% aqueous mobile phase was required to give retention to the polar analyte. This required a column that was designed to work under these conditions and the method included a relatively long equilibrium time between samples.
Literature number: AN796