Feb 2, 2023 1:39:37 PM
Getting started with Flash Chromatography for peptide purification - Tips and Tricks
By Elizabeth Denton

Have you ever tried a new-to-you technique, only to find yourself frustrated by the lack of results? Frustration that only seems to compound when you search for resources that may help you get started and only to come up empty-handed? Me too!! And often for me, this effort ends with me back to using whatever the previous strategy may have been, because whether it's efficient or not, it's the devil I already know.
I've been working with flash chromatography as my primary tool for peptide purification for a couple of years now and honestly encountered some of these similar experiences. But rather than give up, I pushed on and with the help of some other scientists, have identified some strategies that can help make this technique much easier.
In today's post, I've compiled a few of the most helpful strategies into a tips and tricks list that should help you to be more successful using flash chromatography for your peptide purification.
Often times, when learning a new technique or applying an old technique to a new application, there are hurdles that must be overcome and an evolution of mindset that may be required for success. This is absolutely true when you apply flash chromatography, traditionally a tool for small molecule purification, to the purification of peptides. The purity demands can certainly be higher when purifying peptides, but that doesn't immediately eliminate the potential for flash purification in this area of science. To help you get started, here are my top 5 tips and tricks for successfully purifying peptides using flash chromatography:
-
ALWAYS run an analytical HPLC of your crude sample
There are a couple reasons for this, but most importantly the information that is gleaned from even a quick uPLC analytical can inform the design of your actual purification gradient. One critical feature that isn't often discussed is that the acetonitrile concentration required to elute your peptide from the analytical C18 column will remain consistent when you switch over to your flash C18 column.
-
Design a gradient that complements your sample elution profile
Once you have the acetontrile concentration required to elute your peptide you can design a gradient. As a beginner, this can be the most challenging component of preparation for purification because some of the strategies you have learned to improve your HPLC-mediated purification will not help here. For my first injection of a sample, I usually use a gradient that starts 15-20% acetonitrile below the elution concentration and increases linearly about 50% over 10 column volumes before stepping up to 100% acetontrile to flush the column, Figure 1. I have found this starting point works pretty well for most samples.
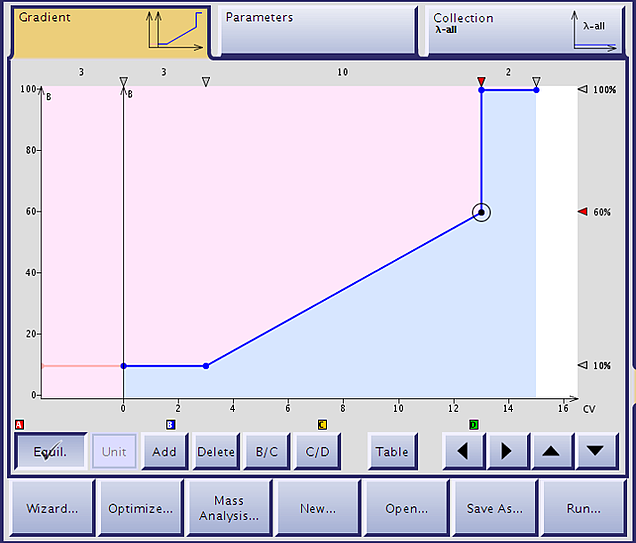
Figure 1. Gradient used to purify a peptide that elutes 25-30% acetontirile as determined with the analytical HPLC. The column is equilibrated at 10% acetonitrile (pink) the sample loaded onto the column (vertical line) the column maintained at 10% to ensure complete removal of the injection solvent, and then purified with a linear gradient increasing a total of 50% acetontrile over 10 column volumes.
-
Use higher concentrations of your mobile phase modifier
Typically, the mobile phase is modified with about 0.1% acid (TFA or formic) to improve the resolution of the peptide(s) in your crude sample. However, given the quantity of material that can be purified on a relatively small flash column, you may need to increase this concentration to ensure all components are fully protonated as desired. I have found that the chromatogram sharpens significantly when you increase the mobile phase modifier concentration from 0.1% up to 0.5% TFA (or other modifiers as well), Figure 2. Sharper peaks also simplifies the follow-up analysis.
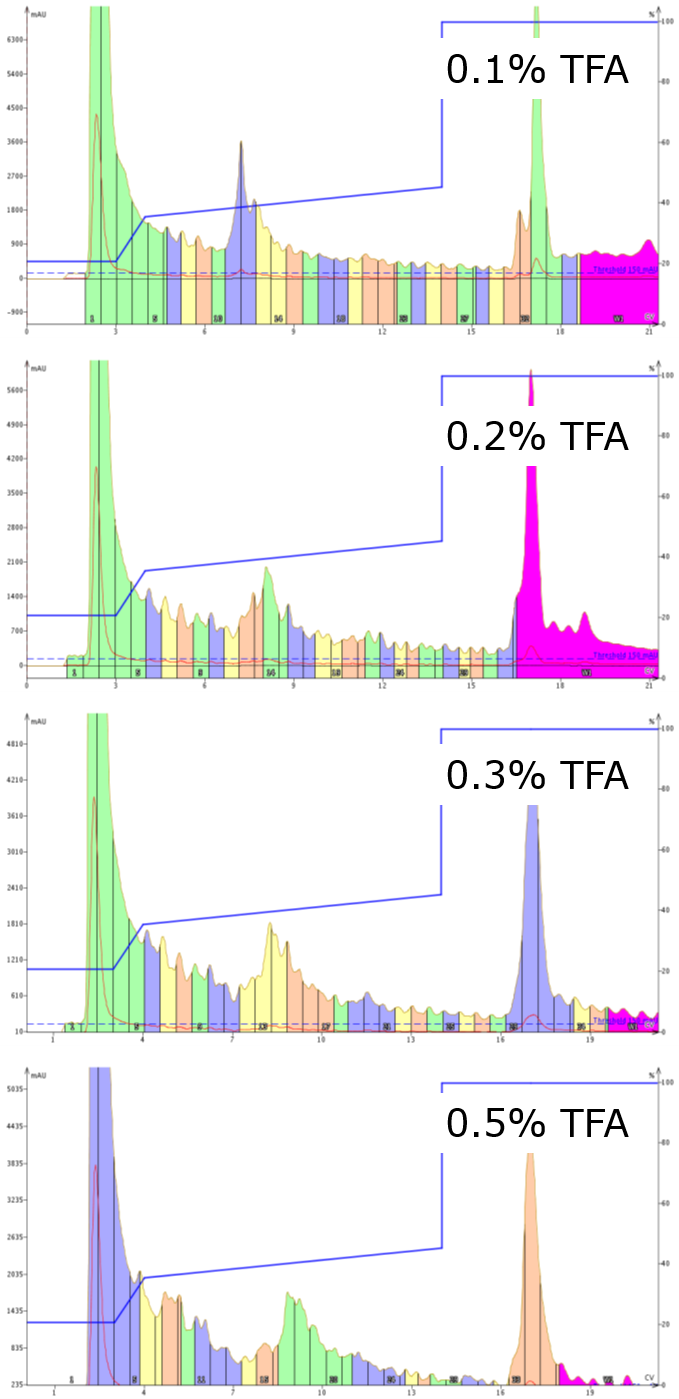
Figure 2. Purification of a 37 amino acid peptide using increasing concentrations of TFA in the mobile phase. Increasing the TFA concentration sharpens the peaks significantly, increasing the probability that you will select the correct fractions with the highest purity and decrease the follow-up analysis.
-
Inject your sample onto the column in as small of a volume as possible
When performing reversed-phase chromatography (of any type really), peak broadening is a more significant concern than if you were performing normal phase chromatography. This is simply due to the partitioning mechanism that is occurring while the sample is on the column. To minimize peak broadening, I recommend to dissolve your sample in as small of a volume as possible for loading onto the column. More often than not, I actually use DMSO to dissolve my sample. Using DMSO keeps the sample highly concentrated and doesn't impact the chromatographic outcome.
-
Consider the physiochemical properties of the peptides
Often times, we resort to a "default" set of conditions. HPLC with 0.1% TFA as the modifier, for example. But there maybe a time that this simply doesn't work. If you take into consideration the physiochemical properties of the peptide BEFORE you start, this headache could be avoided entirely. I typically check the isoelectric point of the peptide, in combination with the crude HPLC to determine whether the pH of the mobile phase should be acidic or basic for the best purification outcome. You'd be surprised how dramatic the results could be, Figure 3.
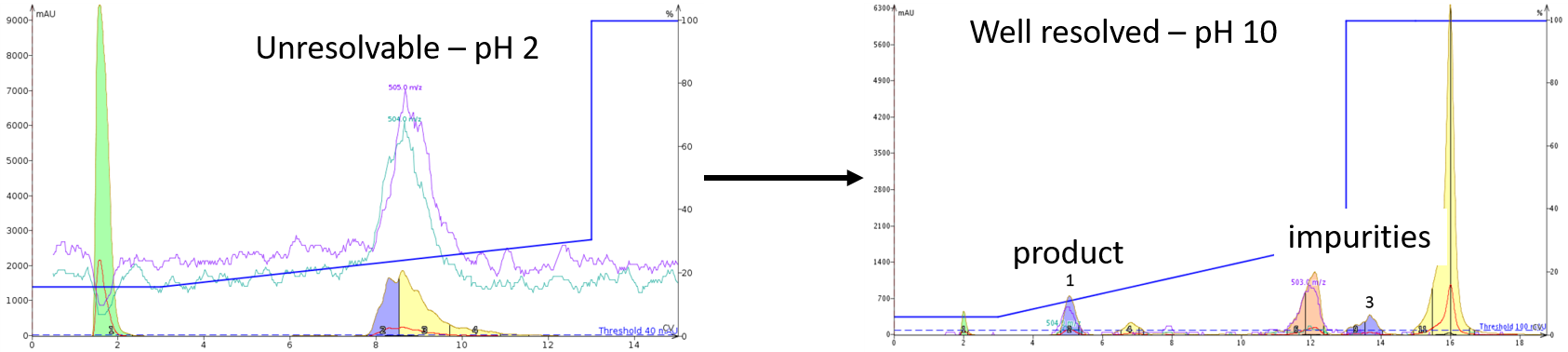
Figure 3. Purification efficiency changes observed by simply changing the mobile phase pH. A sample of oxytocin containing both linear and cyclized peptide was unresolvable at pH 2 (0.1% TFA) but was readily purifiable at pH 10 (0.1% ammonium hydroxide).
Interested in learning more about how techniques that can improve your final peptide purity using flash chromatography? Follow the link below.

