Jan 31, 2023 2:55:42 PM
Increasing your peptide purity using a focused gradient with flash chromatography
By Elizabeth Denton

Reversed phase flash chromatography is increasingly being utilized by peptide chemists to decrease purification time and efforts. The larger particles used in flash columns enable large crude sample loads and can lead to highly pure peptide samples despite lower resolution when compared to traditional HPLC methods. However, there are some situations where the purity achieved isn't sufficient. Then what can you do?
In today's post, I'll describe using a focused gradient to achieve higher purity peptides than is possible with a more traditional linear gradient.
If there's anything constant about peptides, it's their unpredictability. Whether it's a synthesis that has gone totally sideways, or that one sequence that you just can't dissolve once it's cleaved from the resin, or worse yet that one peptide you can't quite get pure using a standard gradient.
In my experience, most groups start peptide purification with a standard gradient - whether that be a 2% per minute change in acetonitrile or a 1% per minute change in acetonitrile or something else, there has to be a starting point. Once this baseline is established, the gradient can be modified to enable purification of the peptide of interest. The same general protocol is also employed when using reversed-phase flash chromatography for peptide purification. So how do you change the gradient to increase the peptide's purity?
One thing is for sure, decreasing the slope of the gradient and extending the purification time doesn't work well in reversed-phase flash chromatography. Rather than increase resolution between peaks, as is often observed with HPLC, this strategy causes severe peak broadening and loss of peak differentiation and detectability. So if extending the gradient doesn't work well, what other strategies can be used?
I have written some about using a step gradient as a method to overcome the peak broadening issue mentioned above while enabling separation of single amino acid deletion sequences. However, this method can be difficult without some additional software help. As an alternative to a step gradient, a focused gradient could be used.
I first purified a sample of an amphipathic peptide 18A synthesized with my Biotage® Initiator+ Alstra™ using a "standard" linear gradient, Figure 1.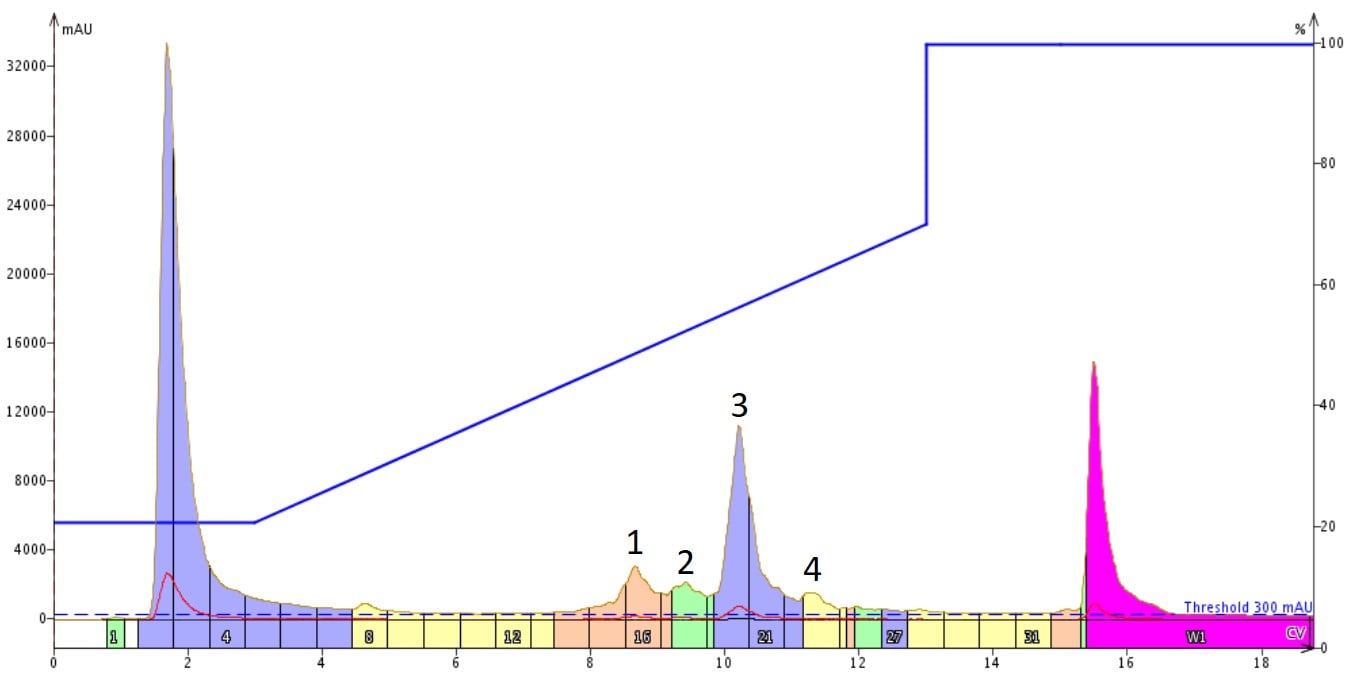
Figure 1. Purification of 18A using a standard linear gradient. The linear gradient has a total of 50% change in acetonitrile (20-70%) over 10 column volumes. The desired peptide is contained in the peak labeled 3.
My standard starting point for a gradient is a 50% change in acetonitrile concentration over 10 column volumes. Most of the time, this approach delivers sufficiently pure product without too much peak broadening, as was the case here. We see the product peak (labeled 3) and three impurity peaks reasonably well defined and separated from each other. After combining fractions and evaporating though, the analytical HPLC indicates that there is still some room for purity improvement, Figure 2.
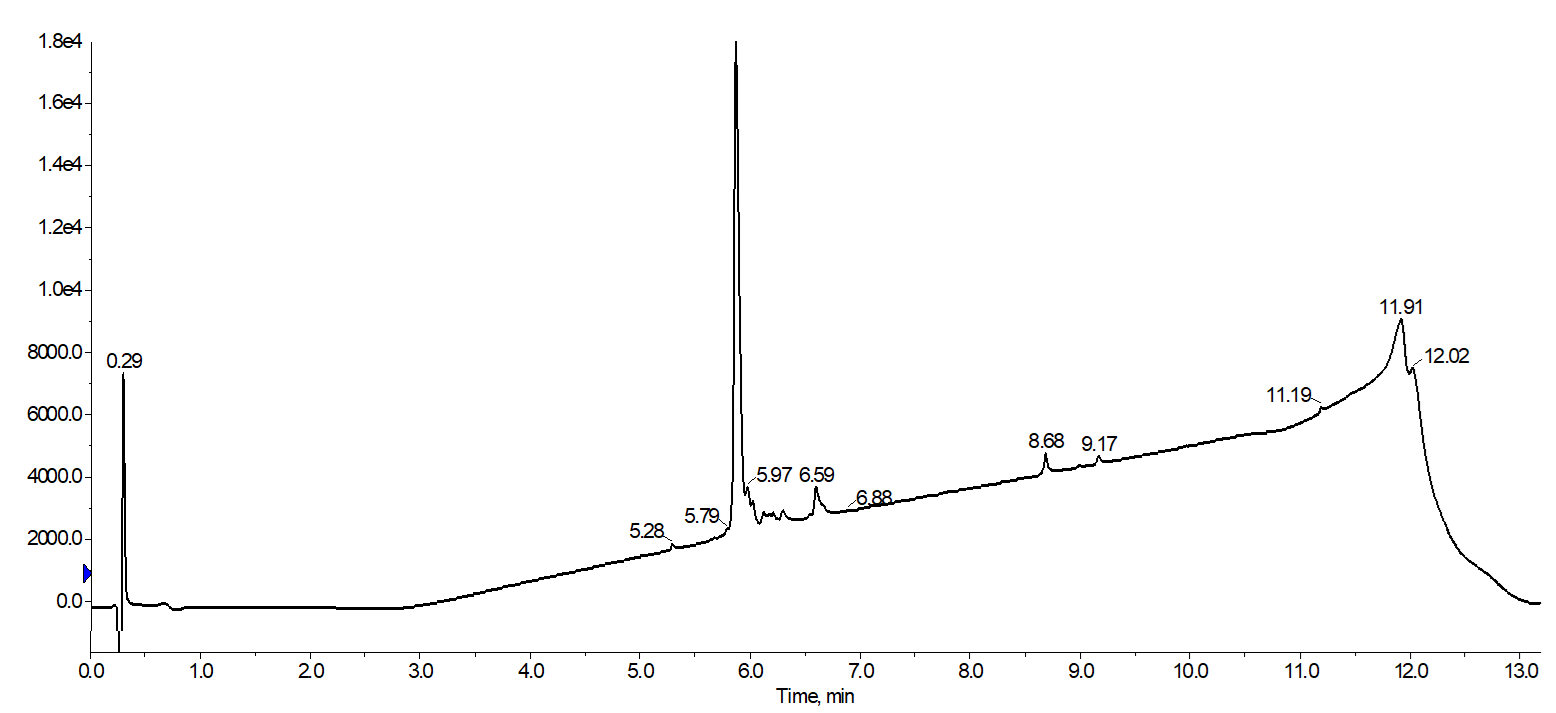
Figure 2. Analytical HPLC of combined 18A fractions collected during purification with a focused gradient. The purification yielded a final product with 83% purity.
I decided to start by approximating the acetonitrile concentration at which the desired peak begins to elute. With this information, I constructed a much more focused gradient. A focused gradient can simply be described as a gradient with regions of sharp changes in mobile phase B concentrations and regions with a very shallow slope near where the desired peptide elutes - in this case surrounding approximately 55% acetonitrile.
I decided to focus the gradient from 50% to 60% acetonitrile, Figure 3. Although this is very shallow when compared to the linear gradient I originally used (1% per CV vs 5% per CV), I hypothesized that the shortness of this shallow gradient would prevent the peak broadening that I have observed previously.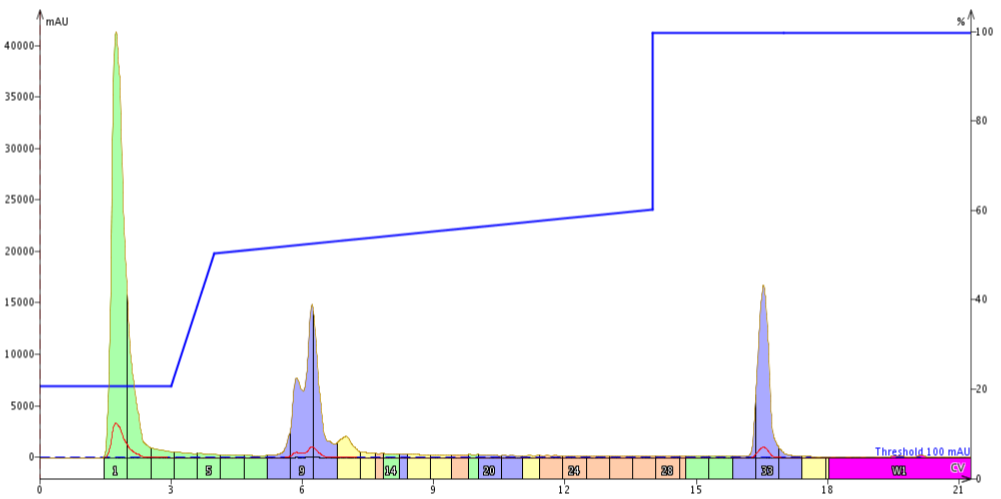
Figure 3. Purification of 18A using a focused gradient across 50-60% acetonitrile. The focused region begins with a concentration that is too high for the early eluting impurities, causing co-elution with the desired product.
Unfortunately, this first try didn't work out as well as I had hoped. Because this gradient was purely focused on the desired peptide, without consideration for the earlier eluting impurities, these three peaks essentially merged together. I didn't even bother combining these fractions and evaluating their final purity.
Given the previous result, I decided to try a gradient with a lower starting point. The desired peptide clearly elutes above 50% acetonitrile, but I thought with a shallow gradient maybe it would elute a little earlier while increasing the separation from the early eluting impurities. With this hypothesis, I built the gradient to run from 40% to 50% acetonitrile over 10 column volumes, Figure 4.
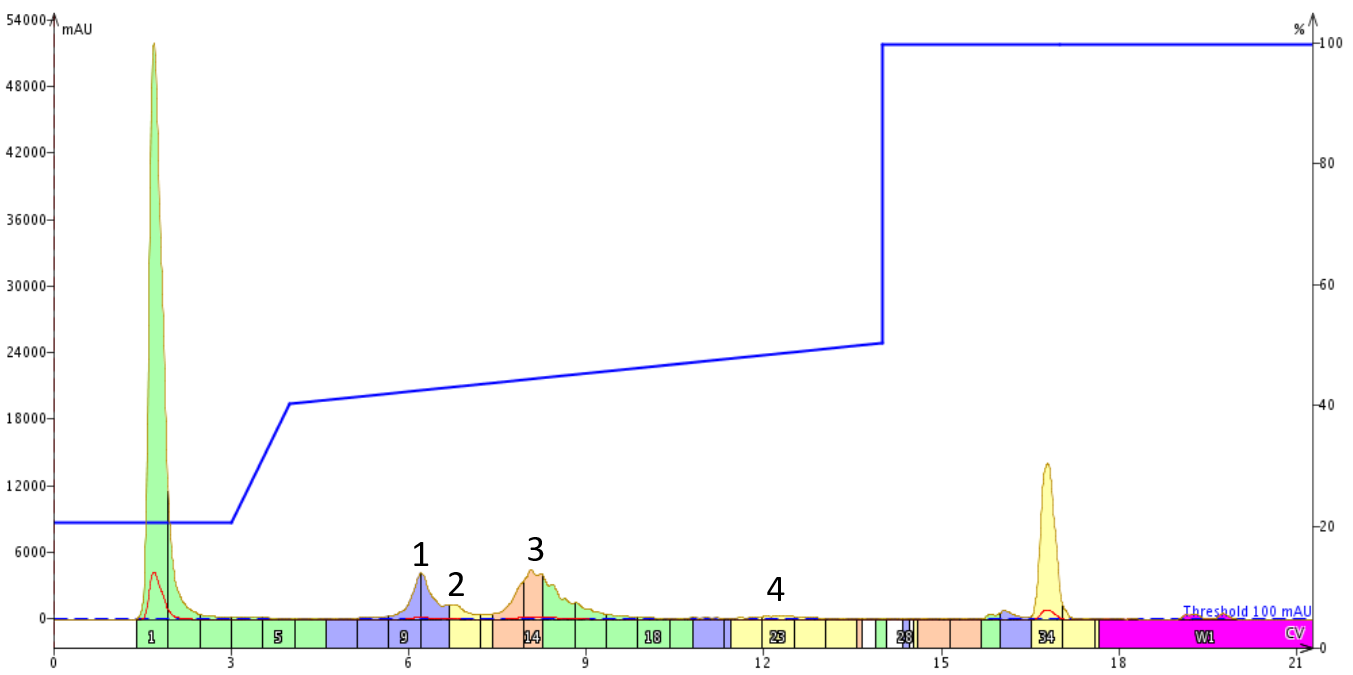
Figure 4. Purification of 18A using a focused gradient with a lower starting point. The three impurity peaks are well resolved from the desired product peak, 3, with minimal peak broadening.
Although the slope of the gradient was the same as the previous purification, simply starting the focused region at a lower acetonitrile concentration improved the resolution between the desired product peak (labeled 3) and the three significant impurities. It is important to note that as the gradient proceeds through the focused region, the later eluting peaks exhibit increasing peak broadening.
I was happy to find after combining fractions 13-15 of the desired product peak and evaporating that the final purity of the sample improved when compared to the "standard" linear gradient, Figure 5.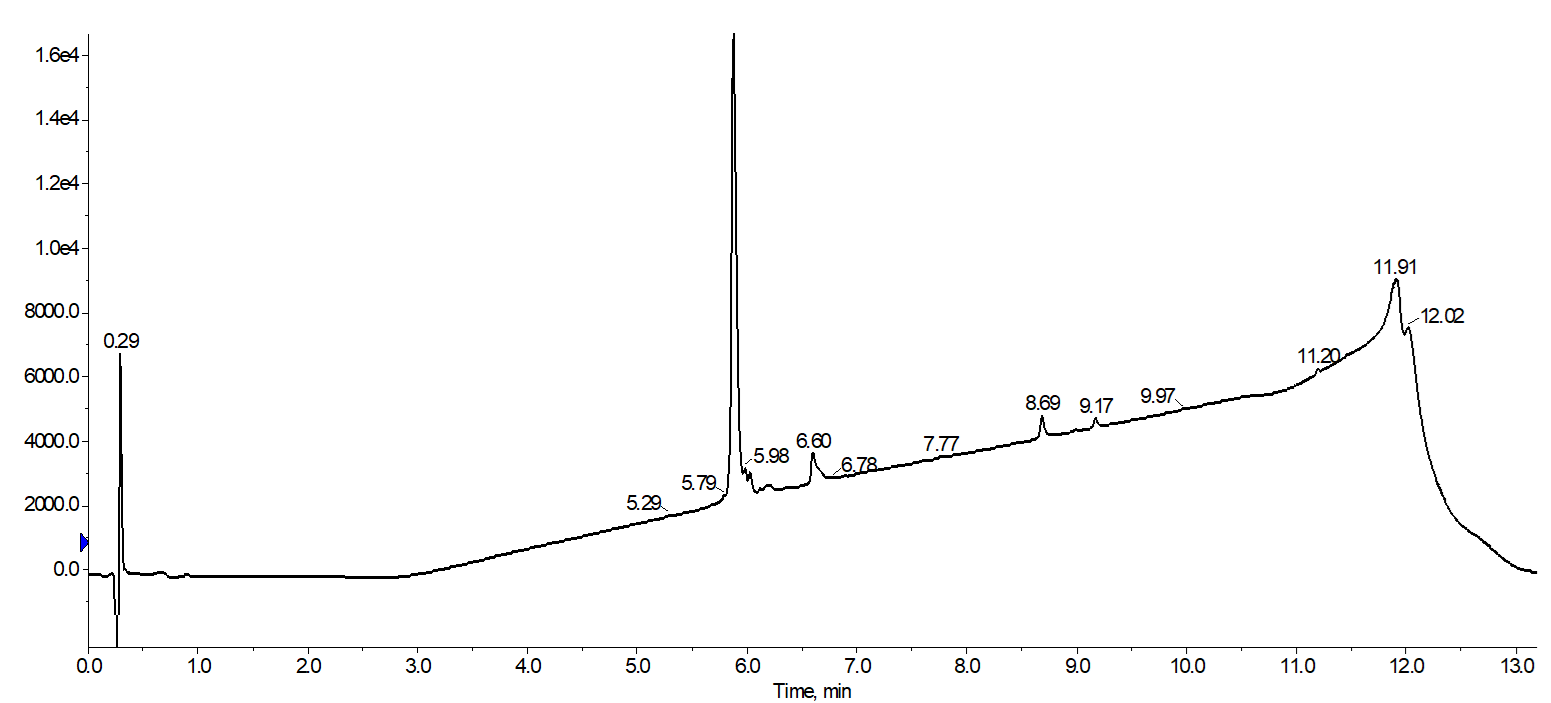
Figure 5. Analytical HPLC of combined 18A fractions collected during purification with a focused gradient. The purification yielded a final product with 88% purity.
For future purifications using a focused gradient, the observed peak broadening will be an important consideration. To avoid significant peak broadening as the purification proceeds, I could envision programming separate regions of focused shallow gradients, particularly if the major impurities elute prior to the desired peptide. Stay tuned for additional work in this area!
Interested in learning more about how flash chromatography can reduce your peptide purification time and efforts? Follow the link below.


