Automated extraction of amphetamines from human urine using ISOLUTE® HCX with Biotage® Extrahera™ HV-5000
For Research Use Only. NOT for Use in Diagnostic Procedures.
 Figure 1. Structures of analytes.
Figure 1. Structures of analytes.
Introduction
This application note describes the extraction of Amphetamines from human urine using ISOLUTE® HCX SPE cartridges and Biotage® Extrahera™ HV-5000 prior to GC/MS analysis.
The simple sample preparation procedure, based on a mixed- mode/strong cation exchange extraction mechanism, delivers clean extracts and analyte recoveries greater than 90% with RSDs lower than 10%. Linearity of greater than 0.998 is achieved from 5–500 ng/mL.
This application note includes optimized conditions for automated processing of the ISOLUTE® HCX cartridges (using Biotage® Extrahera™ HV-5000) and manual processing (using the Biotage® PRESSURE+ 48 positive pressure manifold). Data generated using both processing systems is shown.
Analytes
Amphetamine
Methamphetamine
3,4-Methylenedioxyamphetamine (MDA)
3,4-Methylenedioxymethamphetamine (MDMA)
Methyldiethanolamine (MDEA)
Internal standard
Amphetamine-D5
Sample preparation procedure
Format:
ISOLUTE® HCX 130 mg/3 mL cartridge, p/n 902-0013-B
Automated and manual processing:
Samples were processed manually (batch size 24) using a Biotage® PRESSURE+ 48 positive pressure manifold. Each step below was processed at 2 to 4 psi using the adjustable flow setting. Drying steps were processed at 40 psi using the maximum flow setting.
Automated sample processing (batch size 24) was performed using the Biotage® Extrahera™ HV-5000 system. Detailed processing conditions are included in the appendix.
Sample pre-treatment:
Spike human urine (1 mL) with internal standard solution, add 3 mL 100 mM ammonium acetate (pH6) and mix thoroughly, giving a total volume of 4 mL. Sample pre-treatment is performed in sample tubes (16 x 100 mm, p/n C40708).
Note: Internal standard solution consists of a 10 ng/µL methanolic solution. 10 µL of this was added to 1000 µL of human urine to give a 100 ng/mL spike concentration
Conditioning:
Condition cartridges with methanol (3 mL)
Equilibration:
Equilibrate cartridges with deionized water (3 mL) followed by sodium phosphate buffer (100 mM , pH 6, 1 mL).
Sample loading:
Load the full volume (4 mL) of the pre-treated human urine sample. (This will require two transfer steps as the volume of sample exceeds the volume of the cartridge).
Note: for a smaller batch size of 12, ISOLUTE® HCX 130 mg/10 mL XL (p/n 902-0013-H) cartridges may be used. These facilitate loading of a single transfer step/ aliquot.
Wash 1:
Elute interferences with DI water (2 mL)
Wash 2:
Elute interferences with acetic acid (100 mM, 1 mL)
Wash 3:
Elute interferences with methanol (3 mL)
Dry:
Dry cartridges for 5 mins. under positive pressure (40 psi)
Elution:
Elute analytes with DCM:IPA:NH4OH (78:20:2, 3 mL) into elution solvent tubes (12 x 75 mm, p/n C44651) held in a 24 Position Collection Rack, 12 x 75 mm, p/n 414511SP).
Evaporation and derivatization steps were necessary in this application to obtain a stabilized analyte that could be analyzed via GC/MS.
Post elution and reconstitution:
Transfer elution solvent tubes to the evaporation system.
Evaporation 1: Dry the extract in a stream of air using a TurboVap® 96 Dual at 25 °C, 25 L/min for approximately 55 minutes (note: samples must be completely dry for a successful derivatization step).
Reconstitute evaporated samples with ethyl acetate (50 µL) and PFPA (50 µL). Vortex mix and derivatize on a heat block at 50° for 25 minutes.
Evaporation 2: Transfer to high recovery GC/MS vials and dry in a stream of air using a TurboVap® 96 Dual at 25 °C, 25 L/min for approximately 5 minutes. Recon in 200 µL ethyl acetate and transfer to the GC/MS for analysis.
Evaporation conditions
Evaporation 1
Instrument:
TurboVap® 96 Dual with 24 position rack (12 x 32 mm)
Mode:
Manual
Configuration:
24, single
Gas temperature:
25 oC
Plate temperature:
25 oC
Gas flow:
25 L/min
Plate height:
25 mm
Evaporation 2
Instrument:
TurboVap® 96 Dual with 24 position rack (12 x 32 mm)
Mode:
Manual
Configuration:
24, single
Gas temperature:
25 oC
Plate temperature:
25 oC
Gas flow:
25 L/min
Plate height:
58 mm
GC conditions
Instrument:
Agilent 7890A GC with purged Ultimate Union
Column:
Agilent J&W DB-5ms 30 m, 0.25 mm, 0.25 µm
Mobile phase:
GC/MS grade helium
Inlet temperature:
250 °C Splitless
Flow rate:
1.5 mL/min.
Injection volume:
1.0 μL
Table 1. GC oven gradient.
|
Ramp rate (°C/min) |
Temp (°C) |
Hold Time |
|
|
60 |
1 |
|
25 |
215 |
4 |
MS conditions
Instrument:
Agilent 5975C
Transfer line temp:
280 °C
Source temp:
230 °C
Quad temp:
150 °C
Table 2. MS conditions for target analytes.
|
Analytes |
Quant ions |
Qual ions |
|
Amphetamine |
190 |
118 |
|
Amphetamine-D |
123 |
96 |
|
Methamphetamine |
204 |
118, 160 |
|
MDA |
162 |
135 |
|
MDMA |
162 |
204 |
|
MDEA |
162 |
192 |
Results
Recovery and reproducibility
High (> 90%, figure 2) and reproducible (RSD < 10%, figure 3) recoveries were achieved for all analytes using the method described in this application note using the ISOLUTE® HCX cartridge format processed using the Biotage® Extrahera™ HV-5000. Figure 2. Shows analyte % recoveries from eight spiked samples using the optimized ISOLUTE® HCX protocol described in this application note. Cartridges were processed manually using Biotage® Pressure +48 and extraction was automated using the Biotage® Extrahera™ HV-5000.
Figure 2. Shows analyte % recoveries from eight spiked samples using the optimized ISOLUTE® HCX protocol described in this application note. Cartridges were processed manually using Biotage® Pressure +48 and extraction was automated using the Biotage® Extrahera™ HV-5000. Figure 3. Shows analyte % RSD obtained from eight spiked samples using the optimized ISOLUTE® HCX protocol described in this application note. Cartridges were processed manually using Biotage® Pressure +48 and extraction was automated using the Biotage® Extrahera™ HV-5000.
Figure 3. Shows analyte % RSD obtained from eight spiked samples using the optimized ISOLUTE® HCX protocol described in this application note. Cartridges were processed manually using Biotage® Pressure +48 and extraction was automated using the Biotage® Extrahera™ HV-5000.
Linearity
Calibration curve performance was investigated from whole blood spiked between 5–500 ng/mL. Good linearity was observed delivering r2 values greater than 0.998. Tables 3 and 4. detail linearity performance for each method of extraction.
Table 3. Analyte calibration curve r2 for manual extraction.
|
Analyte |
r2 |
|
Amphetamine |
0.999 |
|
Methamphetamine |
0.998 |
|
MDA |
0.999 |
|
MDMA |
0.999 |
|
MDEA |
0.999 |
Table 4. Analyte calibration curve r2 for automated extraction.
|
Analyte |
r2 |
|
Amphetamine |
0.999 |
|
Methamphetamine |
0.999 |
|
MDA |
0.999 |
|
MDMA |
0.999 |
|
MDEA |
0.999 |
Calibration curves
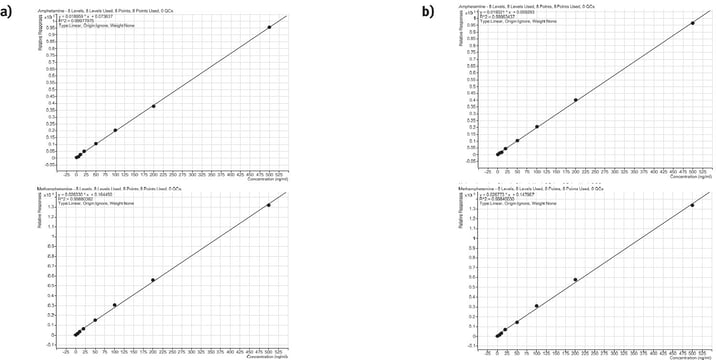
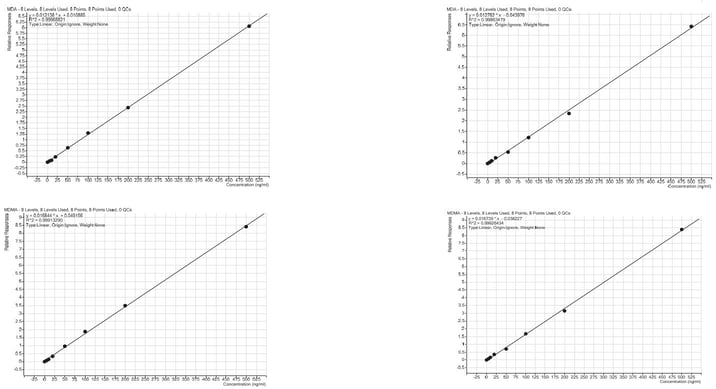
 Figure 4. Calibration curves for automated amphetamine extraction using the Biotage® Extrahera™ HV-5000 (a) and manual extraction (Biotage® PRESSURE+ 48) (b) using ISOLUTE® HCX cartridges.
Figure 4. Calibration curves for automated amphetamine extraction using the Biotage® Extrahera™ HV-5000 (a) and manual extraction (Biotage® PRESSURE+ 48) (b) using ISOLUTE® HCX cartridges.
ISOLUTE® HCX solid phase extraction elution cartridges provided robust automated extraction of amphetamines from pre-treated human urine samples. Good, reproducible recoveries were achieved, with an overall processing time of 58 minutes (excluding the evaporation and derivatisation steps).
Processing time
For a batch size of 24 samples, processing times were:
|
Processing System |
Processing Time |
|
Biotage® PRESSURE+ 48 |
50 minutes |
|
Biotage® Extrahera™ HV-5000 |
58 minutes |
Chemicals and reagents
- Methanol (LC-MS grade), ethyl acetate, IPA and DCM were purchased from Rathburn Chemicals Ltd (Walkerburn, UK).
- All analyte standards, deuterated internal standards, hydrochloric acid, ammonium hydroxide, PFPA and acetic acid were purchased from Sigma- Aldrich Company Ltd. (Gillingham, UK).
- DI Water used was 18.2 MOhm-cm, drawn daily from a Direct-Q5 water purifier.
- Internal standard (100 ng/mL) was prepared from a 1 mg/mL stock solution by adding 10 µL to 990 µL of MeOH. 10 µL of this solution was then added to each calibration solution.
- 100mM Sodium Phosphate (pH6) was made up by adding 1.4196g of sodium phosphate dibasic to 100 mL of water (18.2 MOhm-cm) and adjusting to the correct pH with concentrated hydrochloric acid.
- 100mM ammonium acetate (pH6) was made up by weighing 0.7708g of ammonium acetate, adding 100 mL of water (18.2 MOhm-cm) and adjusting to the correct pH with concentrated hydrochloric acid.
- Acetic acid 100mM was prepared by adding 572 µL of glacial acetic acid to 100 mL of water (18.2 MOhm-cm).
- Elution solvent (DCM:IPA:NH4OH (78:20:2, v/v)) was made up by measuring out 156 mL of DCM and 40 mL of IPA and adding both to a bottle with 4 mL ammonium hydroxide.
Additional information
All data shown in this application note was generated using human urine donated by healthy human volunteers.
Ordering information
|
Part Number |
Description |
Quantity |
|
SPE Consumable |
||
|
902-0013-B |
ISOLUTE® HCX cartridges, 130 mg/3 mL |
50 |
|
Manual Processing |
||
|
PPM-48 |
Biotage® PRESSURE+ 48 Positive Pressure Manifold |
1 |
|
Automated Processing |
||
|
471002 |
Biotage® Extrahera™ HV-5000 |
1 |
|
417610 |
Configuration Kit 24 Positions Dual Flow - HV |
1 |
|
414174SP |
Column Rack 24 x 3 mL |
1 |
|
414511SP |
Collection Rack 12 x 75 mm 24 positions |
1 |
|
414254SP |
Sample Rack 16 x 100 mm, 24 Positions |
1 |
|
C40708 |
Test Tubes (16 x 100 mm) |
1000 |
|
C44651 |
12 x 75 mm Test Tube |
1000 |
|
Evaporation |
||
|
418000 |
TurboVap® 96 Dual |
1 |
|
418312SP |
24 Position Manifold, Kit |
1 |
|
418319SP |
Rack 12 x 32 mm, 24 Positions |
1 |
Appendix: Biotage® Extrahera™ HV-5000 settings
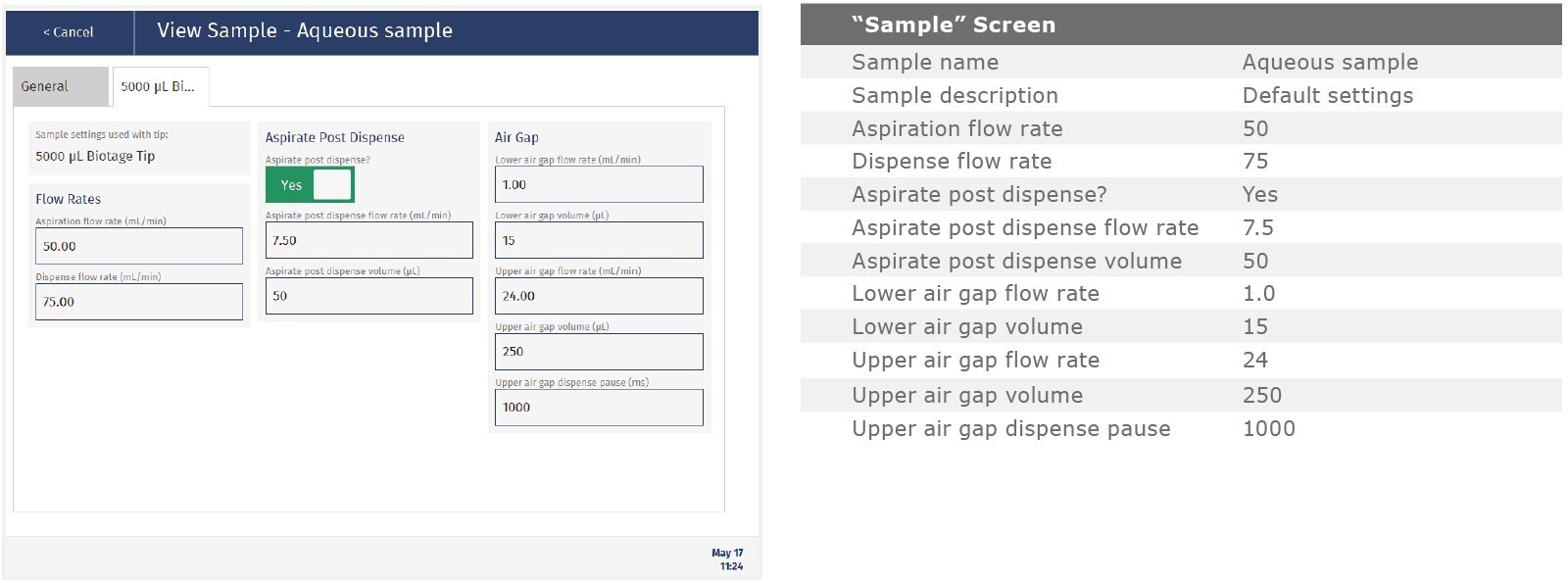
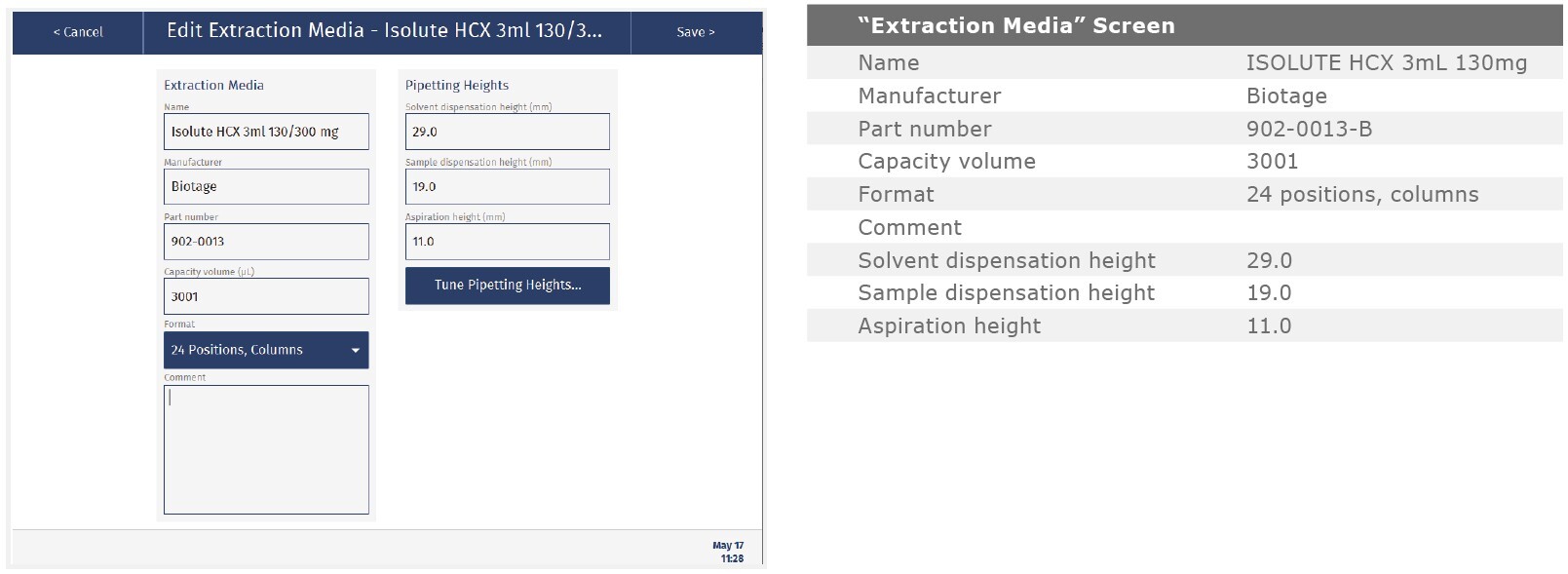
The method described in this application note was automated on the Biotage® Extrahera™ High Volume 5000 using ISOLUTE® HCX 130 mg/3 mL cartridges. This appendix contains the software settings required to configure Extrahera™ to run this method.
Screenshots may not match instrument settings depending upon the instrument software version.
|
Sample name: |
Amphetamines |
|
Sample plate/rack: |
16 x 100 test tubes |
|
Extraction Media: |
ISOLUTE® HCX 130 mg/3mL |
Screenshot Settings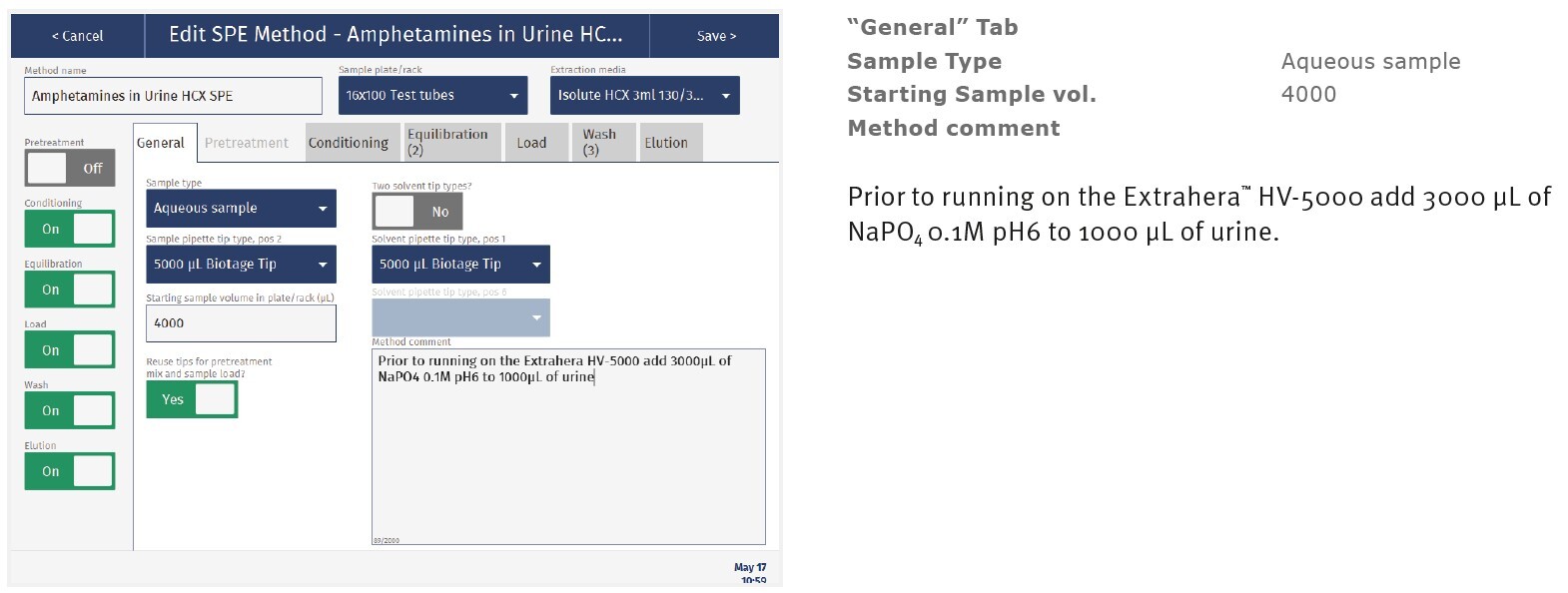
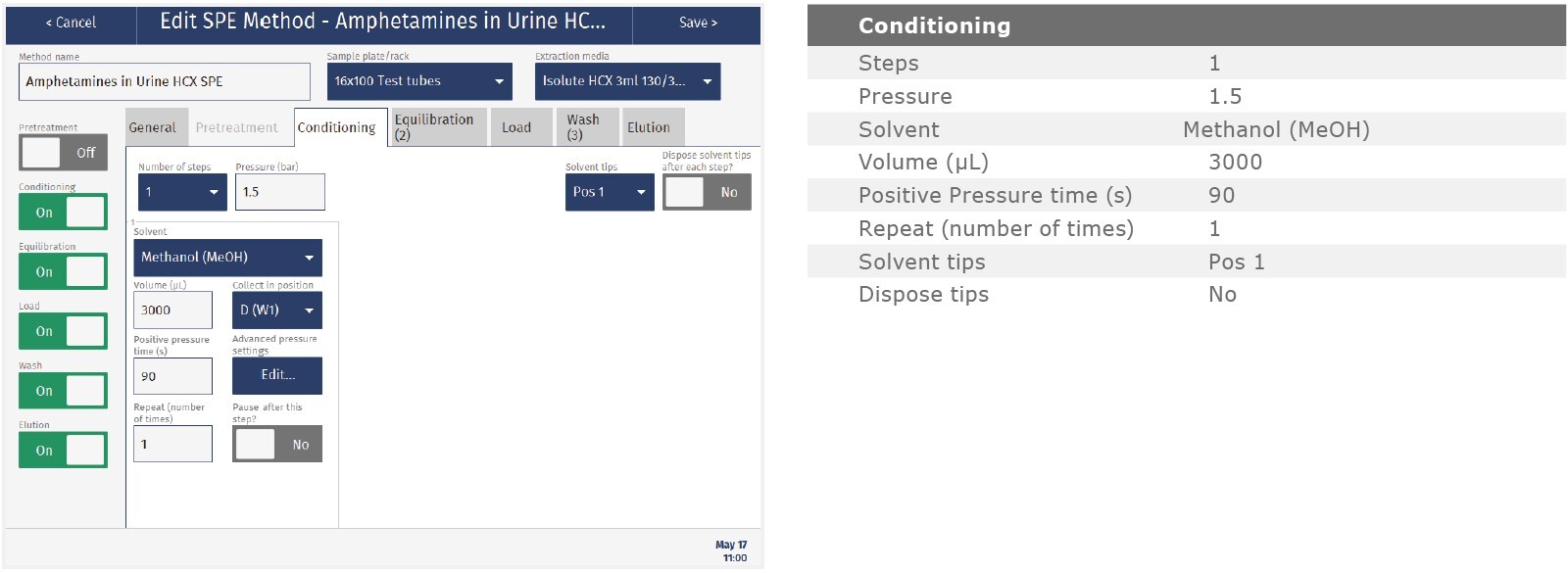
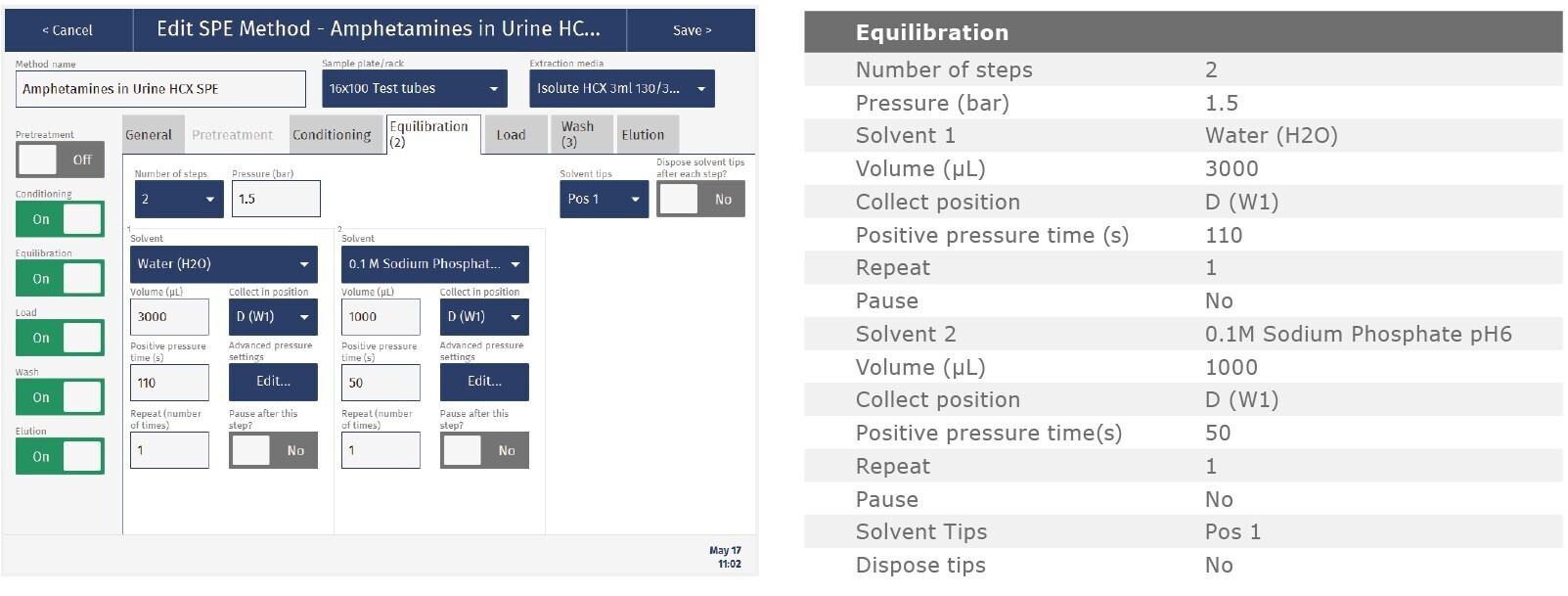
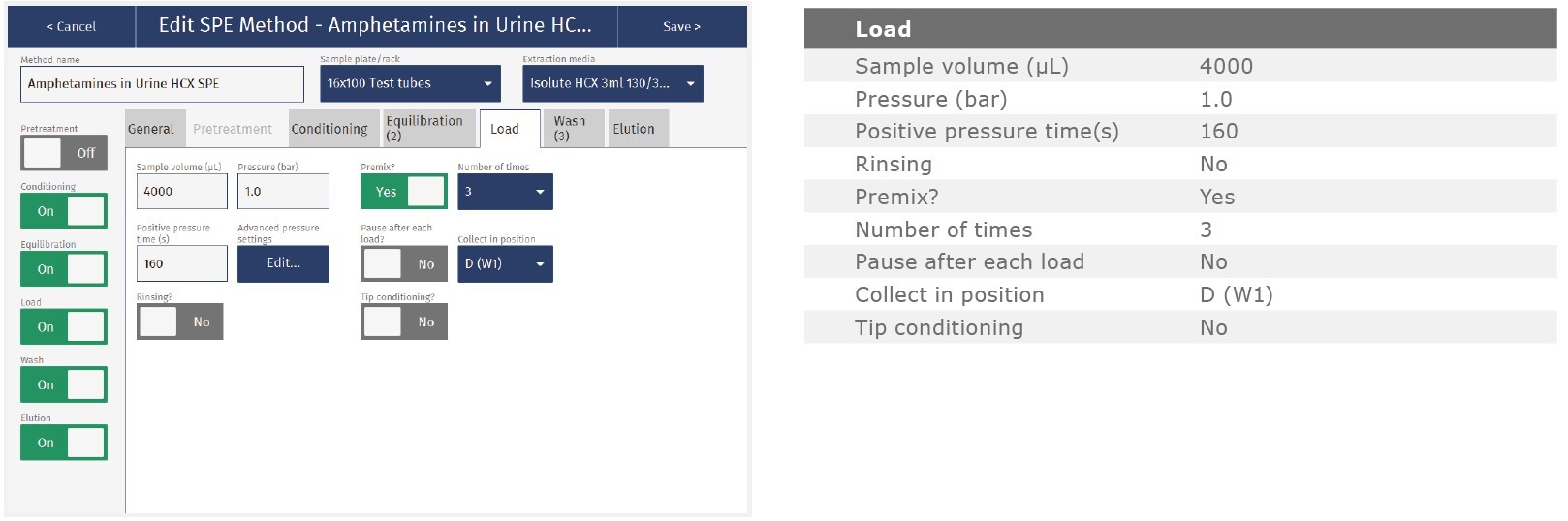
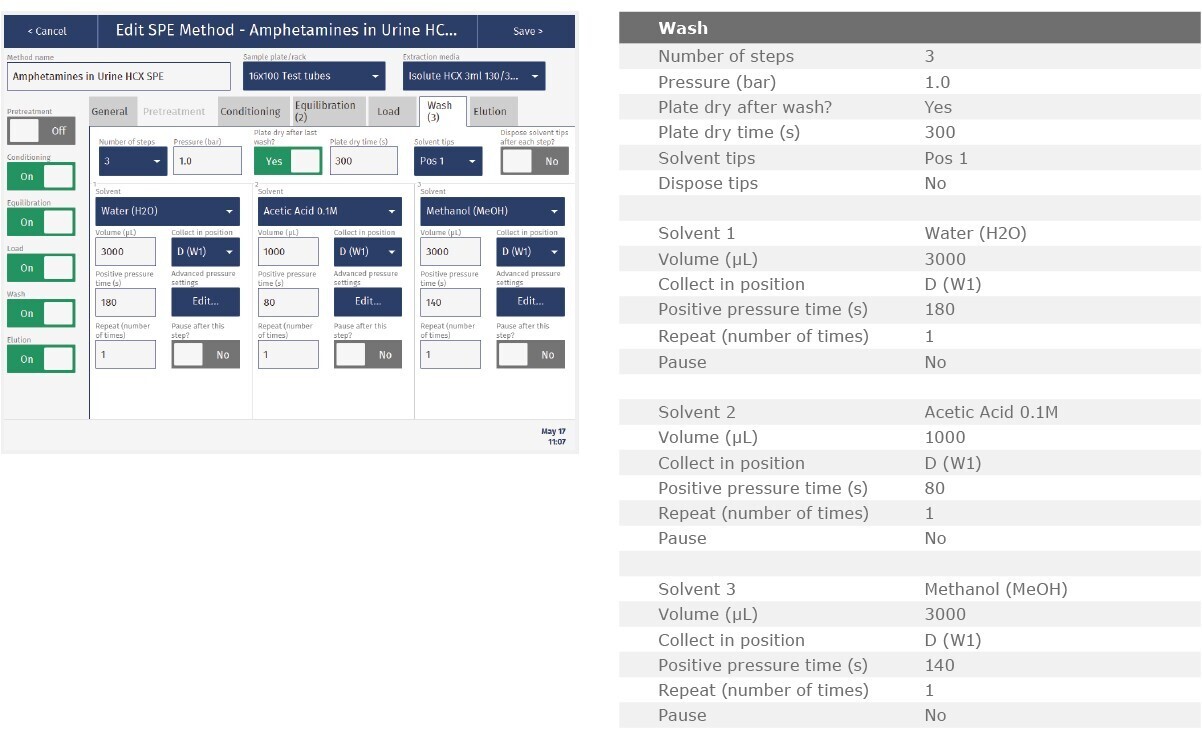
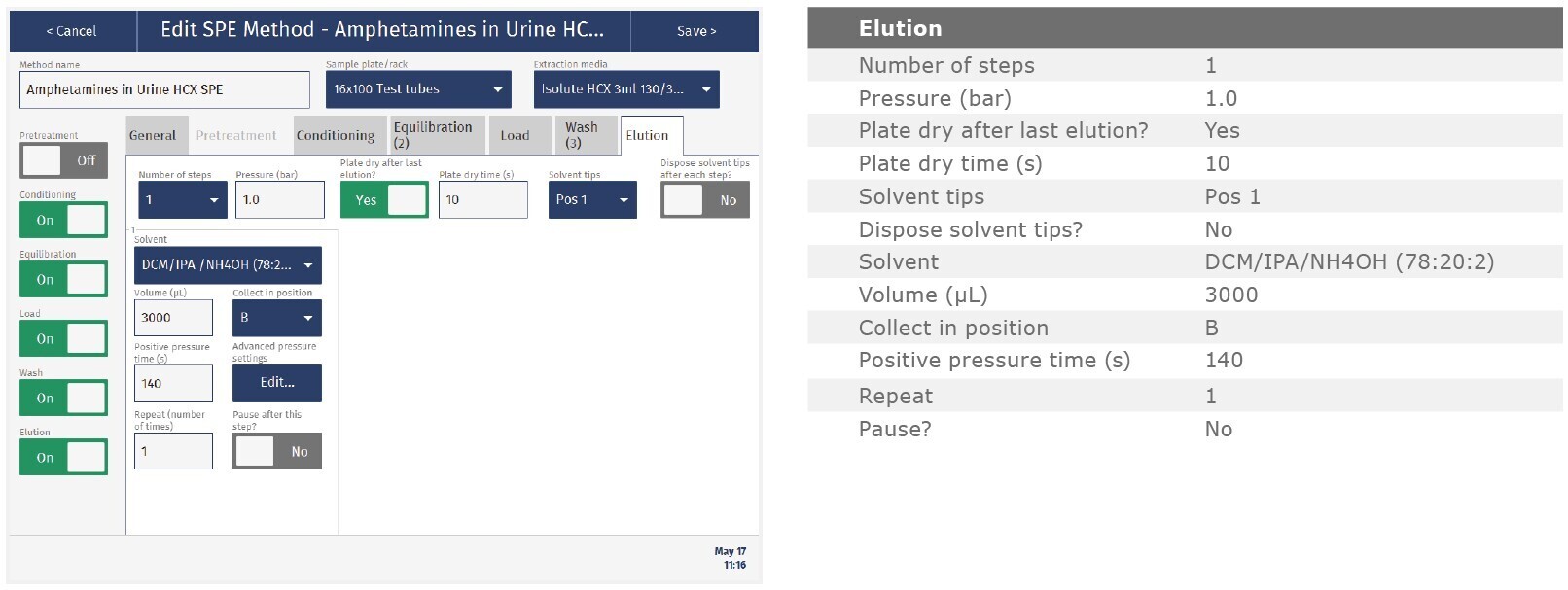
Solvent properties
|
Solvent Description |
|
|
1 |
Methanol |
|
2 |
Water (H2O) |
|
3 |
0.1M Sodium Phosphate, pH6 (aq) |
|
4 |
0.1M Acetic Acid (aq) |
|
5 |
DCM/IPA/NH4OH (78:20:2) |
|
6 |
|
|
7 |
|
|
8 |
|
|
9 |
|
|
10 |
|
|
Solvent |
1 |
2 |
3 |
4 |
5 |
6 |
7 |
8 |
9 |
10 |
|
Reservoir Type |
Refillable |
Non Refillable |
||||||||
|
Capacity |
N/A |
N/A |
N/A |
N/A |
N/A |
|||||
|
Aspiration flow rate |
50 |
50 |
50 |
50 |
50 |
|||||
|
Dispense flow rate |
70 |
75 |
75 |
75 |
40 |
|||||
|
Aspiration post dispense? |
Yes |
Yes |
Yes |
Yes |
Yes |
|||||
|
Aspirate post dispense flow rate |
7.5 |
7.5 |
7.5 |
7.5 |
6.0 |
|||||
|
Aspirate post dispense volume |
50 |
50 |
50 |
50 |
100 |
|||||
|
Lower air gap flow rate |
1.0 |
1.0 |
1.0 |
1.0 |
1.0 |
|||||
|
Lower air gap volume |
15 |
15 |
40 |
40 |
30 |
|||||
|
Upper air gap flow rate |
10 |
24 |
24 |
24 |
5 |
|||||
|
Upper air gap volume |
250 |
250 |
250 |
250 |
250 |
|||||
|
Upper air gap dispense pause |
1000 |
1000 |
1000 |
1000 |
2000 |
|||||
|
Conditioning? |
Yes |
Yes |
Yes |
Yes |
Yes |
|||||
|
Frequency |
1st Asp. only |
|||||||||
|
Cond. Times |
3 |
2 |
2 |
2 |
6 |
|||||
|
Cond. Flow rate |
50 |
50 |
50 |
50 |
75 |
|||||
|
Volume dependent conditioning |
Yes |
Yes |
Yes |
Yes |
Yes |
|||||

Literature Number: AN978

