Automated extraction of a drugs of abuse panel from human urine using Biotage® Extrahera™ LV-200 and micro-elution SPE prior to UPLC-MS/MS analysis
By Biotage
Figure 1. Example analyte structures by drug class.
Introduction
This application note describes the extraction of a multi-class drugs of abuse panel from human urine using Biotage® Mikro CX solid phase extraction micro elution plates, prior to
LC-MS/MS analysis.
The simple sample preparation procedure, based on a mixed- mode/strong cation exchange extraction mechanism, delivers clean extracts and analyte recoveries mostly greater than 60% with RSDs lower than 5% for most analytes. Linearity of greater than 0.999 is achieved for all analytes from 1-1000 pg/mL.
The use of Biotage® Mikro SPE plates for extraction allows for low elution volumes and enhanced workflow efficiency. This application note includes optimized conditions for automated processing of the Mikro plates (using Biotage® Extrahera™ LV-200, see appendix for settings) and manual processing (using the Biotage® PRESSURE+ 96 positive pressure manifold). Data generated using both processing systems is shown. Prior to analysis, extracts are evaporated using the TurboVap® 96 Dual.
Analytes
Amphetamine, Methamphetamine, 3,4-Methylenedioxyamphetamine (MDA), 3,4-Methyl enedioxymethamphetamine (MDMA), 3,4-Methylenedioxy- N-ethylamphetamine (MDEA), Hydromorphone, Morphine, Benzoylecgonine (BZE), Oxymorphone, Dihydrocodeine, Oxycodone, Mephedrone, Norfentanyl, 7-amino-fluni- trazipam, 7-amino-clonazepam, Hydrocodone, Codeine, 6-Monoacetylmorphine (6-MAM), Cocaine, Norketamine, 2-Ethylidene-1,5-dimethyl-3,3-diphenylpyrrolidine (EDDP), Zaleplon, Norbuprenorphine, Ketamine, Nitrazepam, Flunitrazepam, Clonazepam, -OH-triazolam, Oxazepam,
Estazolam, Temazepam, Zolpidem, Alprazolam, Methadone, Lorazepam, Bromazepam, α-OH-alprazolam, 2-OH-ethyl- flurazepam, Triazolam, Nordiazepam, Diazepam, Midazolam, Fentanyl, Flurazepam, Buprenorphine, Phencyclidine (PCP), Lysergic acid diethylamide (LSD).
Internal standards:
Amphetamine-D5, Morphine-D3, BZE-D3, 6-MAM-D3, Diazepam-D5.
Sample preparation procedure
Format:
Biotage® Mikro CX Plate, 2 mg, p/n 601-0002-LVP
Sample pre-treatment:
Spike urine (1 mL) with internal standard solution and allow to equilibrate for 1 hour. Dilute sample with 100 mM NH4OAC pH 5 (950 µL ) and add α-glucuronidase (50 µL). Incubate at 60°C for 2 hours.
Internal standard solution consisted of a 10 pg/ µL methanolic solution. 100 µL of this was added to 1 mL of urine to give a 1 ng/mL spike concentration. Automated and Manual Processing Conditions: Detailed automated processing conditions using the Biotage® Extrahera™ LV-200 system are included in the appendix.
To compare method performance, samples were also processed manually using a Biotage® PRESSURE+ 96 positive pressure manifold. Each step described below was processed at 6 to 9 psi using the adjustable flow setting. Drying steps were processed at 40 psi using the maximum flow setting.
Condition (optional):
Condition wells with methanol (100 µL)
Equilibration (optional):
Equilibrate wells with 4% phosphoric acid (aq) (100 µL)
Sample loading:
Load 400 µL of the pre-treated urine sample
Wash 1:
Elute interferences with 4% phosphoric acid (aq) (100 µL). On completion dry plate for 2 mins.
Wash 2:
Elute interferences with H2O:MeOH (50:50, v/v, 100 µL). On completion dry plate for 2 mins.
Elution:
Elute analytes with DCM:MeOH:NH4OH (78:20:2, v/v, 30 µL) into a 2 mL collection plate (p/n 121-5203)
Post elution & reconstitution:
Dry the extract in a stream of air or nitrogen using a TurboVap® 96 Dual at 25 °C, 60 L/min, plate height 46 mm. Reconstitute evaporated samples with H2O:MeOH (90/10, v/v) containing 0.1% formic acid (30 µL).
Cover with a sealing mat, vortex mix and transfer to 1.5 mL LC/ MS vial with 250 µL glass inserts topped with snap caps (LC/ MS vials: Supelco p/n 854974; Snap Caps: VWR p/n 548-3206; Inserts: Agilent p/n 5183-2085).
UHPLC conditions
Instrument
Shimadzu Nexera UHPLC
Cartridge
Restek Raptor™ Biphenyl 2.7 µm (100 x 2.1 mm) (p/n 9309A12 )
Mobile phase
A: 2 mM ammonium formate (aq) containing 0.1% formic acid
B: 2 mM ammonium formate (MeOH) containing 0.1% formic acid
Flow rate
0.4 mL/min
Injection volume
5 μL
Cartridge temperature
30 °C
|
Time (min) |
%A |
%B |
|
0 |
80 |
20 |
|
2.00 |
80 |
20 |
|
7.50 |
40 |
60 |
|
11.25 |
40 |
60 |
|
12.75 |
0 |
100 |
|
13.50 |
0 |
100 |
|
13.51 |
80 |
20 |
|
15.00 |
80 |
20 |
MS conditions
Instrument:
Shimadzu 8060 Triple Quadrupole MS using ES interface
Nebulizing gas flow:
3 L/min
Drying gas flow:
3 L/min
Heating gas flow:
17 L/min
Interface Temp:
400 °C
DL Temp:
250 °C
Heat block temp:
300 °C
CID gas flow:
270 kPa
|
Analytes |
MRM Transition |
Collision Energy |
Analytes |
MRM Transition |
Collision Energy |
|
Morphine-D3 |
289.0>201.1 (289.0>152.1) |
-26.0 -50.0 |
7-Aminoflunitrazepam |
283.90>135.05 (283.90>227.05) |
-30.0 -26.0 |
|
Morphine |
286.0>152.1 (286.0>201.1) |
-50.0 -25.0 |
Zolpidem |
308.00>235.10 (308.00>263.10) |
-35.0 -25.0 |
|
Oxymorphone |
302.00>227.1 (302.00>198.1) |
-30.0 -45.0 |
Buprenorphine |
468.10>396.25 (468.10>414.30) |
-40.0 -35.0 |
|
Hydromorphone |
286.0>185.0 (286.0>157.0) |
-30.0 -40.0 |
Fentanyl |
337.00>188.10 (337.00>105.00) |
-20.0 -40.0 |
|
Amphetamine-D5 |
141.0>93.0 (141.0>124.15) |
-15.0 -20.0 |
Flurazepam |
388.00>315.00 (388.00>288.00) |
-20.0 -26.0 |
|
Amphetamine |
136>91.05 (136>119.1) |
-15.0 -14.0 |
PCP |
244.00>91.05 (244.00>159.15) |
-35.0 -14.0 |
|
Methamphetamine |
150.0>90.95 (150>119.1) |
-20.0 -14.0 |
Midazolam |
325.90>249.10 (325.90>223.00) |
-35.0 -40.0 |
|
MDA |
180>105 (180>77) |
-20.0 -40.0 |
Bromazepam |
315.80>182.10 (315.80>209.10) |
-31.0 -27.0 |
|
Dihydrocodeine |
302>119.05 (302>171) |
-35.0 -45.0 |
EDDP |
278.00>234.00 (278.00>234.00) |
-30.0 -45.0 |
|
Codeine |
300.0>215.1 (300.0>165) |
-25.0 -40.0 |
Lorazepam |
320.80>275.00 (320.80>229.05) |
-22.0 -30.0 |
|
6-MAM-D3 |
331.0>165.1 (331.0>211.1) |
-40.0 -25.0 |
Oxazepam |
320.80>229.05 (286.90>104.20) |
-23.0 -35.0 |
|
6-MAM |
328.0>165.1 (328.0>211.1) |
-40.0 -25.0 |
Nitrazepam |
286.90>104.20 (281.90>180.10) |
-25.0 -35.0 |
|
MDMA |
194.0>163.1 (194.0>105.0) |
-15.0 -25.0 |
Clonazepam |
315.90>270.05 (315.90>214.05) |
-25.0 -38.0 |
|
Oxycodone |
316.2>241.2 |
-20.0 |
a-OH-Triazolam |
358.90>331.10 (358.90>239.05) |
-28.0 -44.0 |
|
|
178.00>145.05 (178.00>144.00) |
20.0 -30.0 |
|
||
|
Mephedrone |
|
332.90>211.10 (332.90>109.00) |
-37.0 -27.0 |
||
|
|
2-OH-et-flurazepam |
||||
|
|
300.0>199.05 (300.0>171.1) |
-30.0 -40.0 |
|
||
|
Hydrocodone |
Methadrone |
310.50>265.10 |
-16.0 |
||
|
MDEA |
208>163.05 (208>105.05) |
-15.0 -25.0 |
a-OH-Alprazolam |
324.90>216.10 (324.90>205.10) |
-39.0 -46.0 |
|
Nor-ketamine |
223.9>125 (223.9>179.05) |
-20.0 -15.0 |
Nordiazepam |
270.90>140.05 (270.90>208.10) |
-26.0 -28.0 |
|
Nor-fentanyl |
233.0>84.05 (233.0>56.05) |
-20.0 -26.0 |
Zaleplon |
305.90>236.15 (305.90>264.20) |
-28.0 -22.0 |
|
BZE-D3 |
293.00>171.05 (293.00>77.00) |
-20.0 -50.0 |
Flunitrazepam |
313.90>268.10 (313.90>239.10) |
-25.0 -35.0 |
|
BZE |
289.90>168.05 (289.90>105.00) |
-20.0 -30.0 |
Estazolam |
294.90>267.05 (294.90>205.05) |
-20.0 -40.0 |
|
Ketamine |
237.90>125.00 (237.90>207.05) |
-30.0 -14.0 |
Temazepam |
300.90>255.05 (300.90>177.05) |
-20.0 -39.0 |
|
7-Aminoclonazepam |
285.90>222.10 (285.90>121.10) |
-25.0 -29.0 |
Triazolam |
342.90>308.10 (342.90>239.05) |
-27.0 -41.0 |
|
Cocaine |
304.00>182.05 (304.00>82.05) |
-20.0 -30.0 |
Alprazolam |
308.90>281.00 (308.90>205.05) |
-25.0 -40.0 |
|
Norbuprenorphine |
414.00>101.25 (414.00>187.20) |
-39.0 -38.0 |
Diazepam-D5 |
289.90>193.05 (289.90>154.00) |
-32.0 -27.0 |
|
LSD |
323.50>208.10 (323.50>223.25) |
-29.0 -23.0 |
Diazepam |
285.10>193.05 (285.10>154.00) |
-32.0 -27.0 |
Results
Analyte recovery and extraction reproducibility High (mostly > 60%) and very reproducible (RSD < 5%) recoveries were achieved using the method described in this application note. Figure 2 below shows average recoveries (n=7) obtained by manual and automated processing procedures.
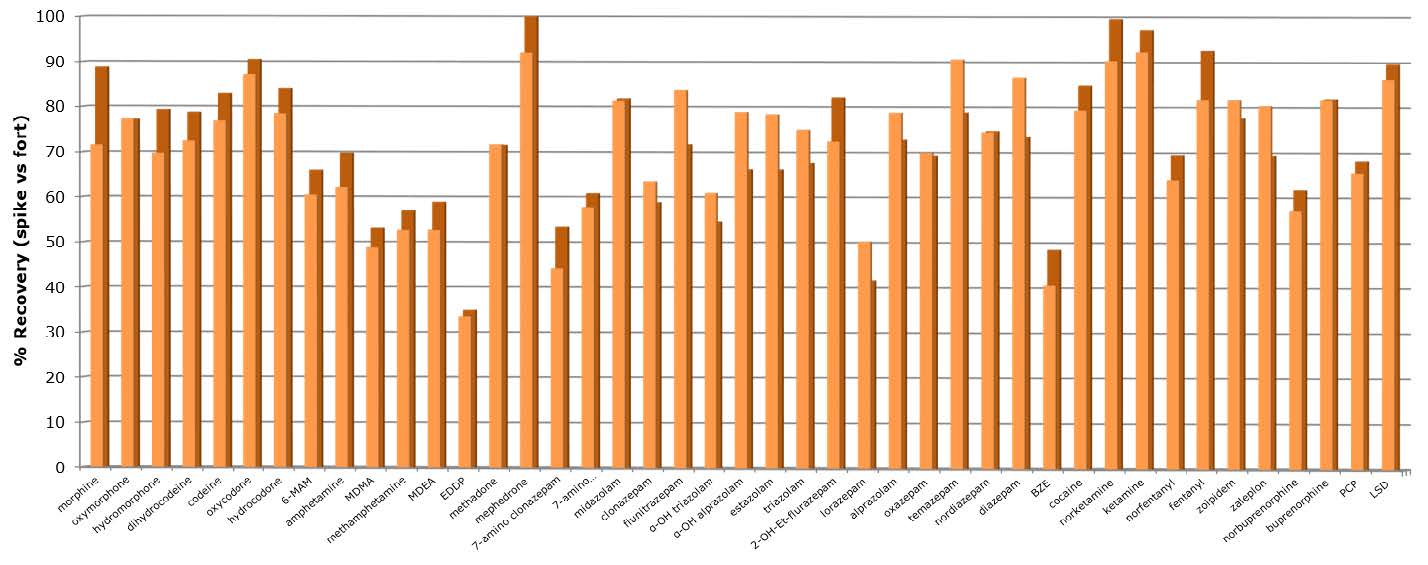
Figure 2. Analyte recoveries (1 ng/mL) using the optimized Biotage® Mikro CX protocol described in this application note. Recovery data comparing manual and automated processing is shown.
Linearity and Limit of Quantitation (LOQ)
Calibration curve performance was investigated from plasma spiked between 1-1000 pg/mL. Good linearity was observed for all analytes typically delivering r2 values greater than 0.999. Table below details linearity performance and associated LOQ for each analyte. Data obtained from manual and automated procedures was comparable.
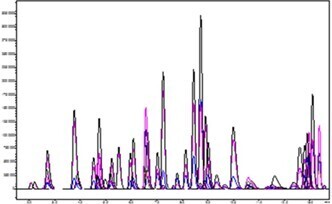
Figure 3. Representative chromatography for application analytes spiked at 1 ng/mL in urine.
|
Analyte |
r2 |
LLOQ (pg/mL) |
Analyte |
r2 |
LLOQ (pg/mL) |
|
Morphine |
0.9997 |
50 |
Buprenorphine |
0.9991 |
25 |
|
Oxymorphone |
0.9991 |
25 |
Fentanyl |
0.9991 |
< 100 |
|
Hydromorphone |
0.9994 |
25 |
Flurazepam |
0.9990 |
5 |
|
Amphetamine |
0.9994 |
50 |
PCP |
0.9992 |
10 |
|
Methamphetamine |
0.9990 |
1 |
Midazolam |
0.9997 |
50 |
|
Dihydrocodeine |
0.9995 |
10 |
Bromazepam |
0.9991 |
50 |
|
Codeine |
0.9996 |
5 |
EDDP |
0.9990 |
1 |
|
6-MAM |
0.9993 |
< 25 |
Lorazepam |
0.9990 |
250 |
|
MDMA |
0.9994 |
10 |
Oxazepam |
0.9990 |
< 500 |
|
Oxycodone |
0.9991 |
25 |
Nitrazepam |
0.9991 |
50 |
|
Mephedrone |
0.9998 |
50 |
Clonazepam |
0.9990 |
< 250 |
|
Hydrocodone |
0.9993 |
50 |
a-OH-Triazolam |
0.9992 |
25 |
|
MDEA |
0.9994 |
10 |
2-OH-et-flurazepam |
0.9998 |
50 |
|
Nor-Ketamine |
0.9992 |
10 |
Methadone |
0.9994 |
10 |
|
Nor-Fentanyl |
0.9990 |
5 |
a-OH-Alprazolam |
0.9993 |
100 |
|
BZE |
0.9997 |
5 |
Nordiazepam |
0.9996 |
50 |
|
Ketamine |
0.9991 |
5 |
Zaleplon |
0.9991 |
25 |
|
7-Aminoclonazepam |
0.9990 |
100 |
Flunitrazepam |
0.9992 |
25 |
|
Cocaine |
0.9992 |
25 |
Estazolam |
0.9994 |
< 25 |
|
Norbuprenorphine |
0.9998 |
250 |
Temazepam |
0.9997 |
< 250 |
|
LSD |
0.9992 |
50 |
Triazolam |
0.9994 |
< 5 |
|
7-Aminoflunitrazepam |
0.9991 |
100 |
Alprazolam |
0.9990 |
25 |
|
Zolpidem |
0.9995 |
5 |
Diazepam |
0.9993 |
25 |
Calibration curves
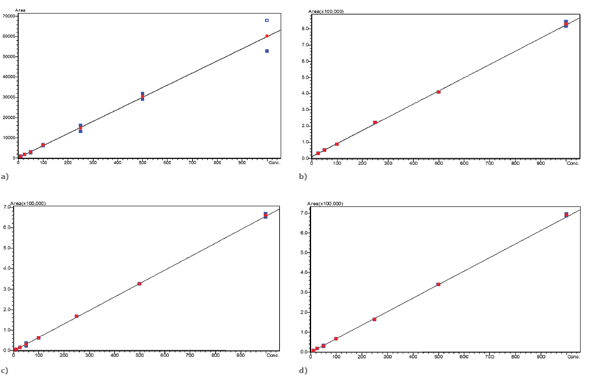
Figure 4. Calibration curves for Burprenorphine (a), Diazepam (b), 6-MAM (c) and Oxycodone (d) using the Biotage® Mikro CX plate to extract hydrolyzed human urine on the Extrahera™ LV-200.
Discussion and conclusion
Biotage® Mikro CX solid phase extraction micro elution plates provided robust automated extraction of a large multi-class drugs of abuse panel from hydrolysed urine samples.
Good, reproducible recoveries were achieved, with an overall automated processing time of ~25 minutes for 96 samples (excluding evaporation and transfer steps). Note: an evaporation step was required in this application, as the elution solvent (DCM/MeOH/NH4OH) which gave the highest analyte recoveries was not compatible with direct injection onto the reversed phase analytical UPLC system. Note: due to the low reconstitution volume used, and issues with compatibility of the available autosampler, reconstituted samples were transferred to low volume inserts prior to injection.
Chemicals and reagents
- Methanol (LC-MS grade), Ultra-Pure Methanol (Gradient MS), and dichloromethane (99.8%) were purchased from Honeywell Research Chemicals (Bucharest, Romania).
- All analyte standards, deuterated internal standards, ammonium acetate, ammonium formate, formic acid, phosphoric acid (49-51%) and ammonium hydroxide (27-30%) were purchased from Sigma- Aldrich Company Ltd. (Gillingham, UK).
- Water used was 18.2 MOhm-cm, drawn daily from a Direct-Q5 water purifier.
- Mobile phase A (2 mM ammonium formate (aq), 0.1% formic acid) was prepared by adding 0.126 mg of ammonium formate to 1 L purified water with 1 mL formic acid.
- Mobile phase B (2 mM ammonium formate (aq), 0.1% formic acid) was prepared by adding 0.126 mg of ammonium formate to 1 L ultra-pure MeOH with 1 mL formic acid.
- Internal standards (100 pg/µL) were prepared from a 10 ng/µL stock solution by adding 10 µL of each of to 950 µL of MeOH. 10 µL of this solution was then added to each calibration solution.
- Hydrolysis buffer 100 mM ammonium acetate was made by adding 0.3854 mg of ammonium acetate to 50 mL of water (18.2 MOhm-cm).
- Equilibration and wash 1 solvent (4% phosphoric
acid) was made by adding 4 mL of phosphoric acid to 96 mL of water (18.2 MOhm-cm). - Wash 2 solvent (H2O:MeOH (50:50, v/v)) was made up by measuring out 50 mL of water (18.2 MOhm-cm) and 50 mL of methanol and adding both to a bottle.
- Elution solvent (DCM:MeOH:ammonium hydroxide (78:20:2, v/v)) was made up by measuring out 78 mL of DCM (18.2 MOhm-cm) and 20 mL of methanol and adding both to a bottle with 2 mL ammonium hydroxide.
- Reconstitution solvent was made by measuring out 90 mL of purified water (18.2 MOhm-cm) and 10 mL of MeOH and adding them to the same bottle with 100 µL formic acid.
Additional information
All data shown in this application note was generated using human urine donated by healthy human volunteers.
Ordering information
|
Part # |
Description |
Quantity |
|
601-0002-LVP |
Biotage® MIKRO CX Plate, 2mg |
1 |
|
121-5203 |
Collection plate, 2 mL, Square |
50 |
|
121-5204 |
Pierceable Sealing Mat |
50 |
|
Automated Processing |
||
|
417000 |
Biotage® Extrahera™ LV-200 |
1 |
|
416920SP |
Pipette Rack, LV/MV |
1 |
|
417423SP |
Pipette Rack, Short |
1 |
|
417008 |
50 µL Clear Tips |
960 |
|
417009 |
200 µL Clear Tips |
960 |
|
Manual Processing |
|
|
|
PPM-96 |
Biotage® PRESSURE+ 96 Positive Pressure Manifold |
1 |
|
Evaporation |
|
|
|
418000 |
TurboVap® 96 Dual |
1 |
Appendix
Biotage® Extrahera™ settings
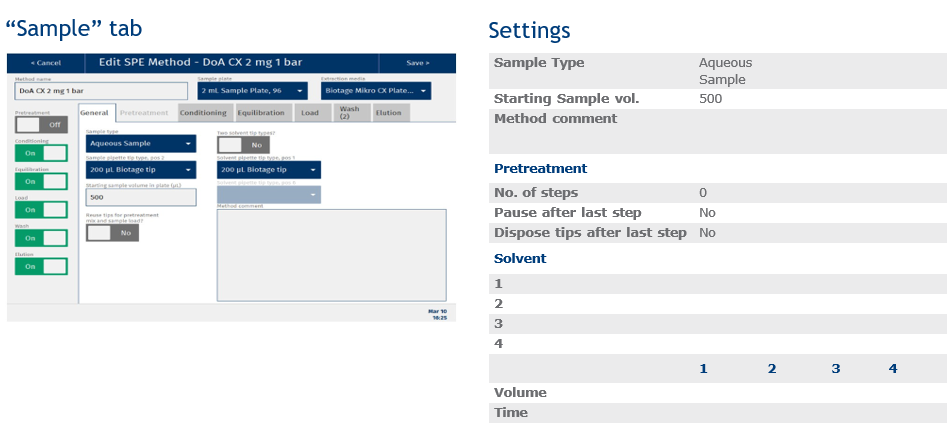
The method described in this application note was automated on the Biotage® Extrahera™ LV-200 using Biotage® Mikro CX plates.
This appendix contains the software settings required to configure Extrahera to run this method. As described in the main body of the application note, analyte recoveries, linearities and LOQs were comparable for both manually processed and automated methods. Reproducibility was slightly improved for samples extracted using the automated Extrahera™ LV-200 system.
Total time for extraction of 96 samples using this method was 25 minutes (excluding pre-extraction sample hydrolysis, and post extraction evaporation and reconstitution time).
|
Sample name: |
DoA CX 2 mg 1 Bar |
|
Sample plate/rack: |
2 mL Sample Plate, 96 |
|
Extraction Media: |
Mikro CX 96 Well Plate |



Solvent properties
|
Solvent description |
|
|
1 |
Methanol |
|
2 |
4% Phosphoric Acid |
|
3 |
DCM/MeOH/NH4OH (78:20:2) |
|
4 |
50:50 Water:Methanol |
|
Solvent |
1 |
2 |
3 |
4 |
5 |
6 |
7 |
8 |
9 |
10 |
|
Reservoir type |
Refillable |
|
|
|
|
Non refillable |
|
|
|
|
|
Capacity |
|
|
|
|
|
|
|
|
|
|
|
Aspiration flow rate |
5 |
1 |
0.6 |
5 |
|
|
|
|
|
|
|
Dispense flow rate |
10 |
10 |
10 |
10 |
|
|
|
|
|
|
|
Lower air gap flow rate |
10 |
10 |
10 |
10 |
|
|
|
|
|
|
|
Lower air gap volume |
5 |
5 |
5 |
5 |
|
|
|
|
|
|
|
Upper air gap flow rate |
10 |
10 |
10 |
10 |
|
|
|
|
|
|
|
Upper air gap volume |
140 |
140 |
50 |
140 |
|
|
|
|
|
|
|
Upper air gap dispense pause |
0 |
0 |
300 |
0 |
|
|
|
|
|
|
|
Conditioning? |
Yes |
Yes |
Yes |
Yes |
|
|
|
|
|
|
|
Cond. Times |
3 |
1 |
4 |
3 |
|
|
|
|
|
|
|
Cond. Flow rate |
5 |
3 |
4 |
5 |
|
|
|
|
|
|
|
Chlorinated |
No |
No |
Yes |
No |
|
|
|
|
|
|
|
Serial dispense |
No |
No |
No |
No |
|
|
|
|
|
|

Literature number: AN964

