Extraction of vitamin D metabolites from human serum using ISOLUTE® SLE+
This application note describes the extraction of Vitamin D metabolites from human serum using ISOLUTE® SLE+ with LC-MS/MS analysis.
Figure 1: Structure of angiotensin II
Introduction
Vitamin D deficiency can result in a variety of health issues such as osteoporosis, liver and kidney problems, it is often associated with increased risk of cancers and multiple sclerosis. From this standpoint vitamin D monitoring is on the rise and of significant clinical relevance. This method describes the use of ISOLUTE® SLE+ plates for the efficient and simple extraction of the vitamin D metabolites 25-OH vitamin D2 and 25-OH vitamin D3. Incorporated into the procedure is an integral protein binding disruption step which maximizes analyte recovery and eliminates the need for any offline protein disruption. This method has been internally validated using DEQAS approved serum samples and deuterated internal standards the results obtained are better than that required for criteria for acceptable performance for DEQAS approval. All recoveries were consistently greater than 90% with all RSDs < 10% at LOQs of 1-3.75 ng/mL.
ISOLUTE® SLE+ Supported Liquid Extraction plates and cartridges offer an efficient alternative to traditional liquid liquid extraction (LLE) for bioanalytical sample preparation, providing high analyte recoveries, no emulsion formation, and significantly reduced sample preparation time.
Analytes
25-OH Vitamin D2 and 25-OH Vitamin D3
ISOLUTE® SLE+ procedure
Format: ISOLUTE® SLE+ 400 Supported Liquid Extraction Plate, part number 820-0400-P01
Sample pre-treatment: Dilute human serum (150 µL) with HPLC grade water: isopropanol (v/v, 50:50) (150 µL ) mix, cap and shake for 60 seconds.
Sample loading: Load pre treated serum (300 µL in total) onto the ISOLUTE® SLE+ plate followed by a pulse of vacuum to initiate flow (VacMaster 96 Sample Processing Manifold 121-9600) , leave the sample to absorb for 5 minutes.
Analyte elution: Apply heptane (750 µL), wait five minutes to allow the solvent to soak, apply a short pulse of vacuum if solvent not fully adsorbed. Apply a second aliquot of heptane (750 µL), allow to soak for a further 5 minutes and then apply a final pulse of vacuum.
Post extraction: Evaporate the eluate to dryness without heat and reconstitute (SPE Dry 96 –SD-9600-DHS-UK) in 100 µL of (v/v, 30:70) 2 mM ammonium formate (aq) with 0.1% formic acid: 2 mM ammonium formate (99% MeOH, 1% aq) with 0.1% formic acid. Cap and vortex gently for 60 seconds.
Post Extraction: Evaporate the eluate to dryness without heat and reconstitute (SPE Dry 96 –SD-9600-DHS-UK) in 100 µL of (v/v, 30:70) 2 mM ammonium formate (aq) with 0.1% formic acid: 2 mM ammonium formate (99% MeOH, 1% aq) with 0.1% formic acid. Cap and vortex gently for 60 seconds.
Additional information: Analytes are particularly sensitive to light. Amber glassware is recommended where possible
HPLC conditions
Instrument: Waters Acquity UPLC fitted with 20 μL Loop.
Cartridge: Restek Pinnacle DB Bi (1.9μ, 50 x 2.1 mm id)
Mobile Phase: A= 2 mM ammonium formate (aq) with 0.1% formic acid, B= 2 mM ammonium formate (99% MeOH 1% aq) with 0.1% formic acid at a flow rate of 0.4 L/min
Injection Volume: 15 μL (partial loop with overfill).
Sample temperature: 10 °C.
Mass spectrometry conditions
Instrument: Quattro Premier XE triple quadrupole mass spectrometer (Waters Assoc., Manchester, UK) equipped with an electrospray interface for mass analysis. Table 1. shows the positive ions acquired in the multiple reaction monitoring (MRM) mode.
Desolvation Temperature: 450 °C
Ion Source Temperature: 150 °C
|
Analyte |
MRM Transition |
Cone Voltage (V) |
Collision Energy (eV) |
|
25-OH Vitamin D2 (Quantification) |
395.5 - 269.5 |
30 |
18 |
|
25-OH Vitamin D2 (Qualification) |
395.5 - 119.2 |
30 |
26 |
|
D6-25-OH Vitamin D3 (Internal Standard) |
389.6 - 263.5 |
30 |
16 |
|
25-OH Vitamin D3 (Quantification) |
383.5 - 257.5 |
30 |
17 |
|
25-OH Vitamin D3 (Qualification) |
383.5 - 107.2 |
30 |
25 |
Results
Optimum extraction conditions were observed using sample pre-treatment dilution 1:1 with 50/50 IPA/H2O. This pretreatment was strong enough to enable protein disruption without effecting full protein precipitation. Validation of this method was carried out using Chromsystems deuterated internal standards over two days. Figures 1 and 2 show the calibration curves constructed using this method as well as the quantification chromatograms for the two Vitamin D analytes. The calibration curves demonstrated excellent linearity with coefficients of determination greater than 0.99 for both analytes. Various DEQAS samples were subsequently run, demonstrating excellent correlation to nominal values supplied by the organization, this data is shown in Table 2. All recoveries were consistently greater than 90% with all RSDs < 10% and well within the standard deviations provided by DEQAS. Limits of quantitation were observed at 3.75 ng/mL for 25-hydroxyvi- tamin-D2 and 1 ng/mL for 25-hydroxyvitamin-D3.
 Figure 2: Chromatograms of 25-hydroxyvitamin D3 and internal standard and extracted chromsystems calibration curve of 25-hydroxyvitamin D3
Figure 2: Chromatograms of 25-hydroxyvitamin D3 and internal standard and extracted chromsystems calibration curve of 25-hydroxyvitamin D3
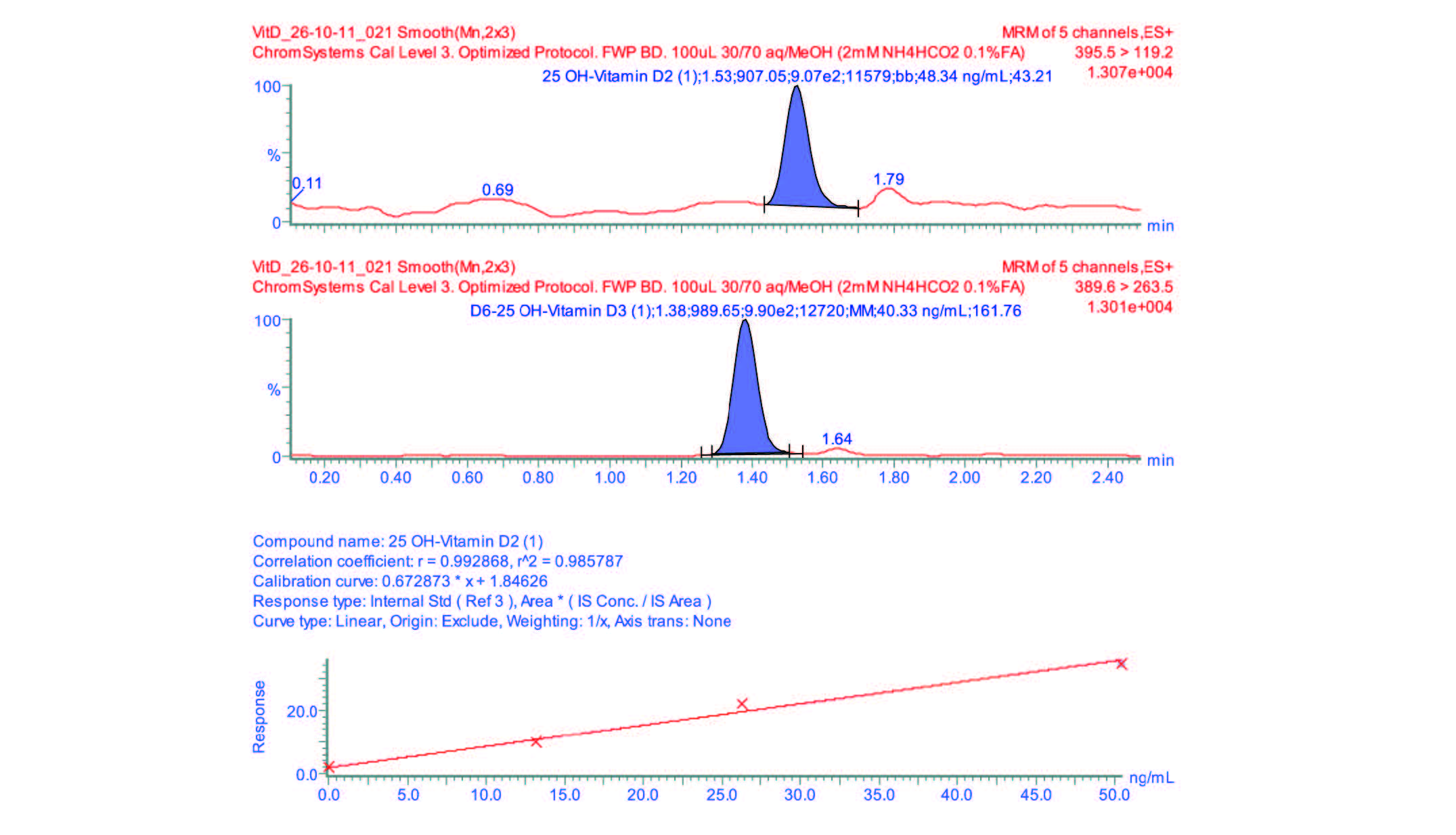
Figure 3: Chromatograms of 25-hydroxyvitamin D2 with internal standard and extracted chromsystems calibration curve of 25-hydroxyvitamin D2
References
All work based on poster presented at MSACL 2012: A Novel Method for the Extraction of 25-Hydroxy-vitamin D and Analysis using UPLC-MS/MS. Rhys Jones, Lee Williams, Helen Lodder, Geoff Davies, Steve Jordan, Claire Desbrow and Gavin Jones. Biotage GB Limited, Distribution Way, Dyffryn Business Park, Ystrad Mynach, Hengoed, CF82 7TS, UK.
|
Sample ID |
DEQAS ALTM using LC/MS |
Low Pass Threshold |
High Pass Threshold |
Biotage Result |
Pass/Fail |
|
DEQAS 381 |
40.2 |
35.1 |
45.3 |
37.5 |
pass |
|
DEQAS 382 |
16.0 |
13.9 |
18.1 |
14.4 |
pass |
|
DEQAS 383 |
33.3 |
30.0 |
37.2 |
31.6 |
pass |
|
DEQAS 384 |
11.8 |
10.2 |
13.4 |
11.3 |
pass |
|
DEQAS 385 |
26.4 |
23.4 |
29.4 |
26.4 |
pass |
Note. In the United States, use of a LC-MS/MS technique to detect underivatized 25-hydroxyvitamin D2 and D3 in a sample using a triple quadrupole instrument in MRM positive-ion mode requires a separate license from the Mayo Foundation for Medical Education and Research under U.S. Patent No. 7,700,365.
Ordering information
|
Part Number |
Description |
Quantity |
|
820-0400-P01 |
ISOLUTE® SLE+ 400 Supported Liquid Extraction Plate |
1 |
|
121-9600 |
Biotage® VacMasterTM-96 Sample Processing Manifold |
1 |
|
|
(without vacum control) |
|
|
SD-9600-DHS-EU |
Biotage® SPE Dry 96 Dual |
1 |
|
|
Sample Concentrator System, 220/240V |
|
|
SD-9600-DHS-NA |
Biotage® SPE Dry 96 Dual |
1 |
|
|
Sample Concentrator System, |
|
Literature number: AN757

