Dec 19, 2023 3:00:00 PM
Are step gradients effective with reversed phase flash chromatography?
By Bob Bickler

With the increasing use of reversed phase flash chromatography in medicinal chemistry and organic synthesis labs comes the desire to speed up and make these purifications more time and solvent efficient. Many of these chemists are familiar with prep HPLC and the long chromatography times this technique often requires, typically 30 + minutes, to complete the purification.
Reversed-phase flash, with its shorter, fatter columns and larger particle size media, can reduce purification times by just following the same HPLC gradient method and operating at faster flow rates. But with time and solvent use a major concern in many labs, any simple way to speed reaction mixture purification without sacrificing purity or yield while reducing solvent consumption at the same time is beneficial.
Step gradients are a chromatographic technique that can reduce purification time and solvent use. There is a caveat – the linear gradient used as the starting point for the step gradient must provide nearly full resolution of the product and its nearest byproducts. Once there is an acceptable linear gradient, then a step gradient can be created through a little trial and error. I have previously posted on how to create a step gradient from a linear gradient.
One of my recent projects synthesized 2,4,6-trichloroaniline. Being lipophilic, this compound and several of the byproducts were best separated using a 60-100% methanol gradient on a C18 reversed-phase flash column, Figure 1.
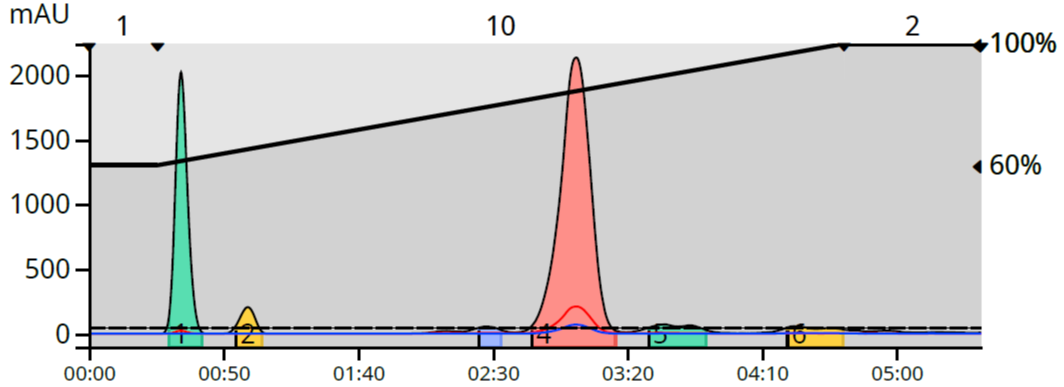 Figure 1. Reaction mixture reversed phase linear gradient.
Figure 1. Reaction mixture reversed phase linear gradient.
As with most linear gradients, this one I designed to run for 10 column volumes (CV) with the addition of a 1 CV hold at 60% methanol at the start of the gradient and another 2 CV hold (100% methanol) at the end for a total of 13 CV to ensure a good separation between the target and its closest eluting byproducts. Total gradient volume using a 12-gram Biotage® Sfär C18 column with this method was 221 mL, and at a flow rate of 40 mL/min, required a little over 5.5 minutes to complete. Pretty darned fast but can be improved upon.
After a little method development, I found that a step gradient starting at 75% methanol (1 CV) followed by a step to 80% methanol (held for 4 CV), and finally a step to 100% (held for 2 CV) created an equal separation but required only 3.7 minutes and consumed only 149 mL, Figure 2.
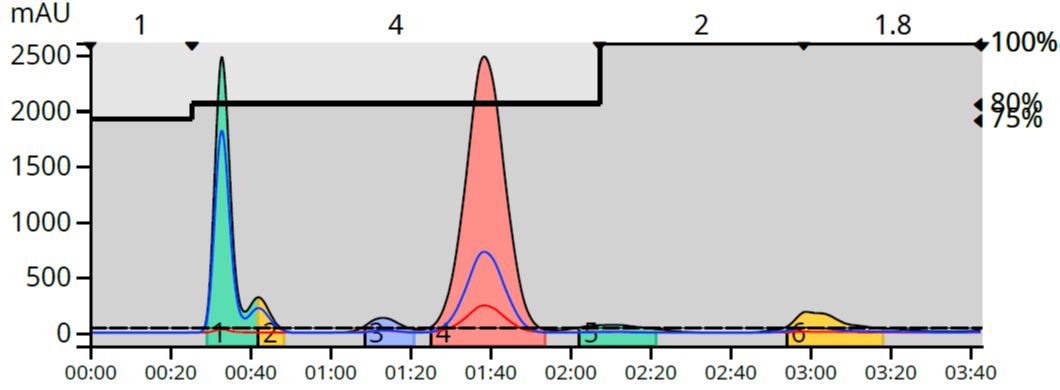 Figure 2. Reaction mixture step gradient purification reduced elution time by 2 minutes compared to a 10 CV linear gradient.
Figure 2. Reaction mixture step gradient purification reduced elution time by 2 minutes compared to a 10 CV linear gradient.
While these time and volume differences do not seem to be that remarkable, just think about the impact the step gradient would have with larger columns. Solvent savings in excess of 38% with a 33% reduction in purification time are achievable when a step gradient is put into use.
You may be wondering how well step gradients handle larger loads compared to linear gradients. I have that data as well. The linear and step gradient methods were compared with 113 mg loads (~1% of media mass) using a Biotage dry load vessel (DLV) for dry loading and the same 12-gram C18 column, Figure 3.
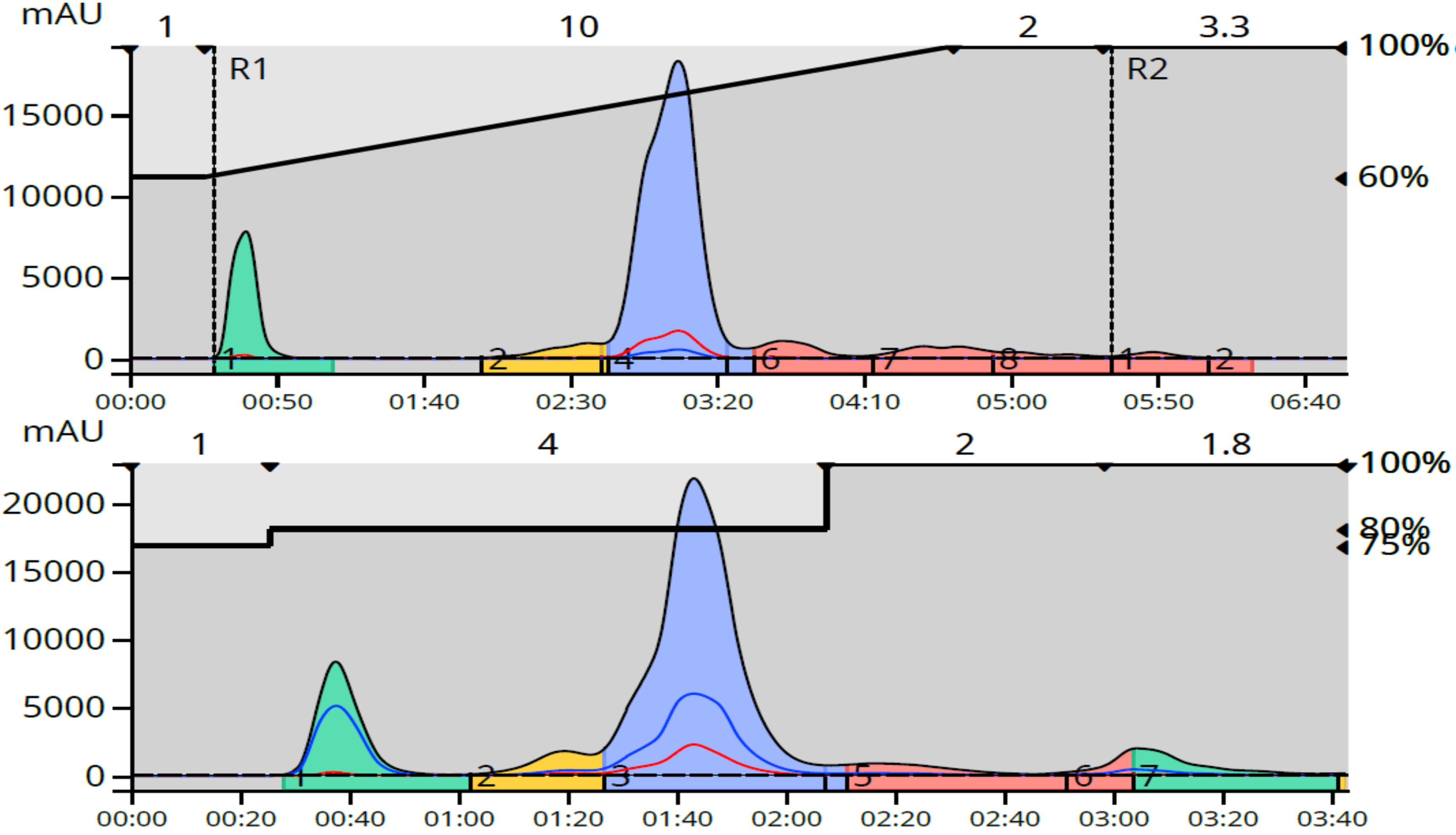 Figure 3. Step gradient chromatography (bottom) purified the reaction mixture three minutes faster than a linear gradient at equivalent loads with similar results.
Figure 3. Step gradient chromatography (bottom) purified the reaction mixture three minutes faster than a linear gradient at equivalent loads with similar results.
While both methods generated similar purification results, the step gradient, as mentioned earlier, required even less time than the linear as many late-eluting compounds were detected at the end of the linear gradient causing the method to continue another 3.3 CV (56 mL).
The product fraction from both purifications were analyzed and found to be of identical purity, with similar recovery, Figure 4.
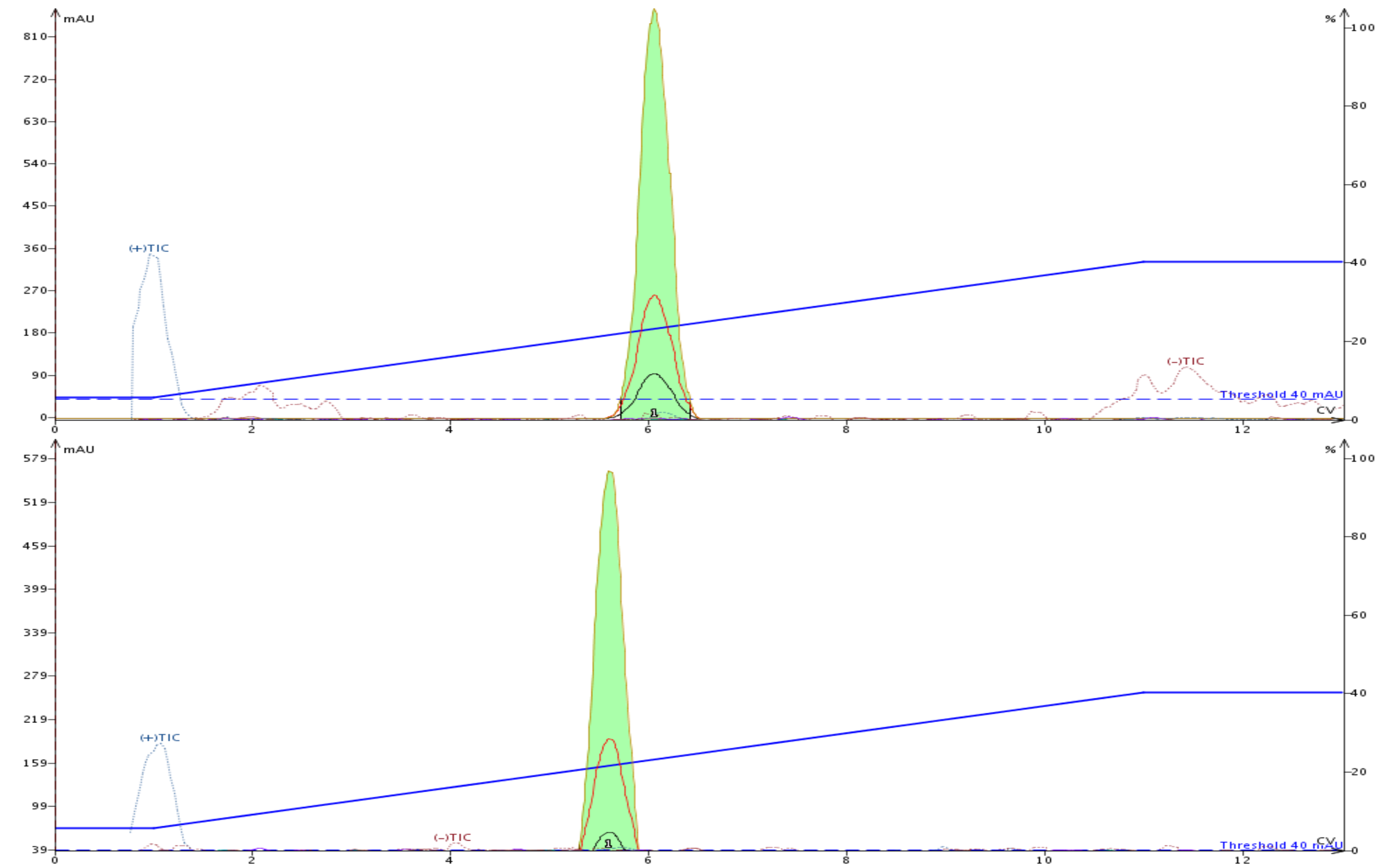 Figure 4. Fraction purity analysis showed similar product purity. Top- linear gradient purified product. Bottom-step gradient purified product.
Figure 4. Fraction purity analysis showed similar product purity. Top- linear gradient purified product. Bottom-step gradient purified product.
So, consider using a step gradient when scaling up a reversed-phase purification. It will increase your throughput and reduce purification costs.
For more information on this topic, download our whitepaper - How to Determine Reversed-phase Flash Chromatography Loading Capacity

