Feb 6, 2024 3:00:00 PM
Can organic solvent choice impact reversed phase flash chromatography separations?
By Bob Bickler

The answer to this question is absolutely yes. The choice of organic solvent can impact your reversed phase flash purification results just as organic solvent selection impacts normal phase results.
Organic solvents fall into different classes – polar, protic, aprotic, non-polar, etc. They also fit into different selectivity groups. Solvents in one selectivity group may separate compounds in a sample in a different order or with different spacing than other solvents.
With reversed-phase flash chromatography, the two organic solvents used most frequently are acetonitrile and methanol. While both are polar, acetonitrile is aprotic and methanol is protic, so there is that difference. They also fall into different selectivity groups. Because of these differences, it is a good idea to test your crude mixture’s chromatography with a water/acetonitrile gradient and a water/methanol gradient to determine which provides the best results for your targeted compound. In some cases, acetonitrile will be better, in others, methanol will prevail.
Let’s look at some examples where the same crude reaction mixture was chromatographed using water/ methanol and water/acetonitrile.
- Natural product extract – Ylang Ylang III.
- This terpenoid containing extract has several compounds of various polarity. Purification with methanol provided a better separation of the more polar terpenoids but not of the more lipophilic compounds, Figure 1.
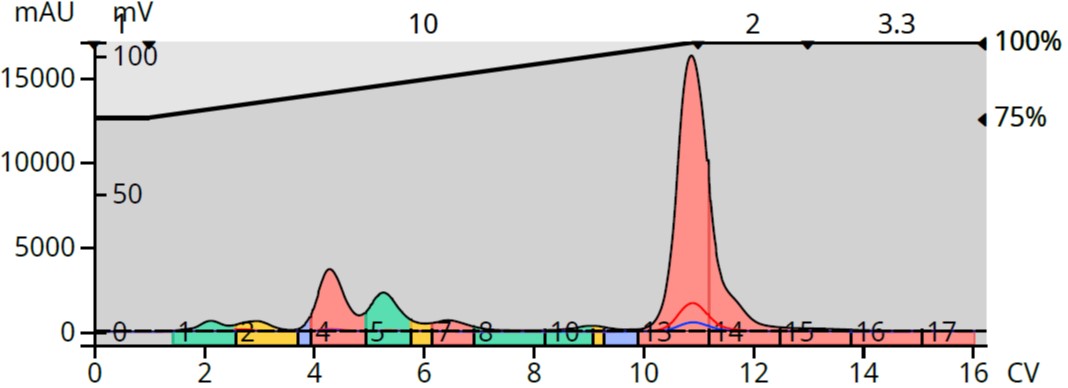
Figure 1. Ylang Ylang III oil chromatography with a 75-100% MeOH in water gradient. the pole polar terpenes are better separated than the later eluting compounds.
Purification using acetonitrile, however, provided a partial separation of the more lipophilic sesquiterpenes but failed to provide an acceptable separation of the early eluting terpenoids, Figure 2.
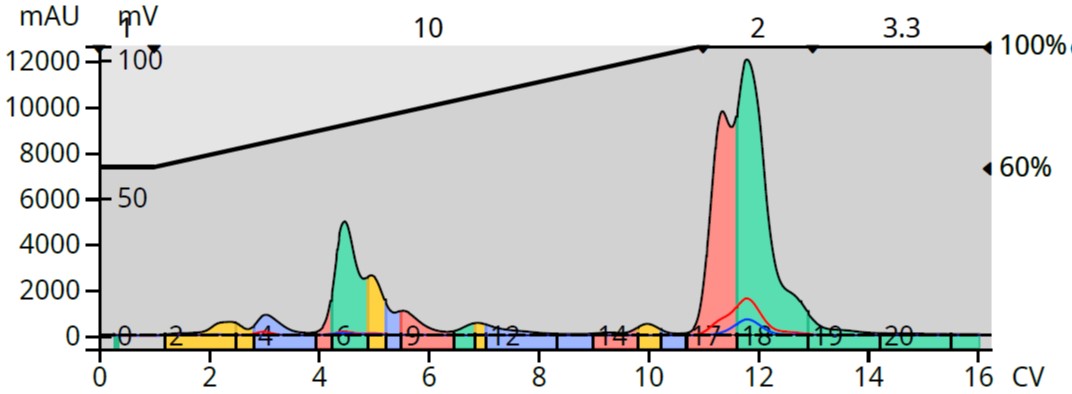
Figure 2. Ylang Ylang III oil chromatography with a 75-100% acetonitrile in water gradient. The more lipophilic terpenes are better resolved than the more polar compounds.
- Synthetic reaction mixture – reaction of isatoic anhydride, nicotinuric acid, α-methylbenzylamine.
- This reaction mixture contains compounds with a broad polarity range including acids, esters, and amides. For this sample, a water/acetonitrile solvent provided the better separation though the methanol purification did elute a pair of lipophilic compounds apparently not soluble in acetonitrile, Figure 3.

Figure 3. Reaction mixture chromatography comparison. Top - water/acetonitrile gradient. Bottom - water/methanol gradient. For this reaction mixture, the water/acetonitrile method is superior.
5-comp mix – naphthalene, 1-nitronaphthalene, 3,5-dibenzyloxyacetophenone, methyl paraben, butyl paraben.
I often use for training purposes as it works for both normal- and reversed phase purifications. Reversed-phase chromatography of this mixture using water/methanol and water/acetonitrile highlights the selectivity, or the spacing differences, between a few pairs of adjacent compounds, Figure 4.
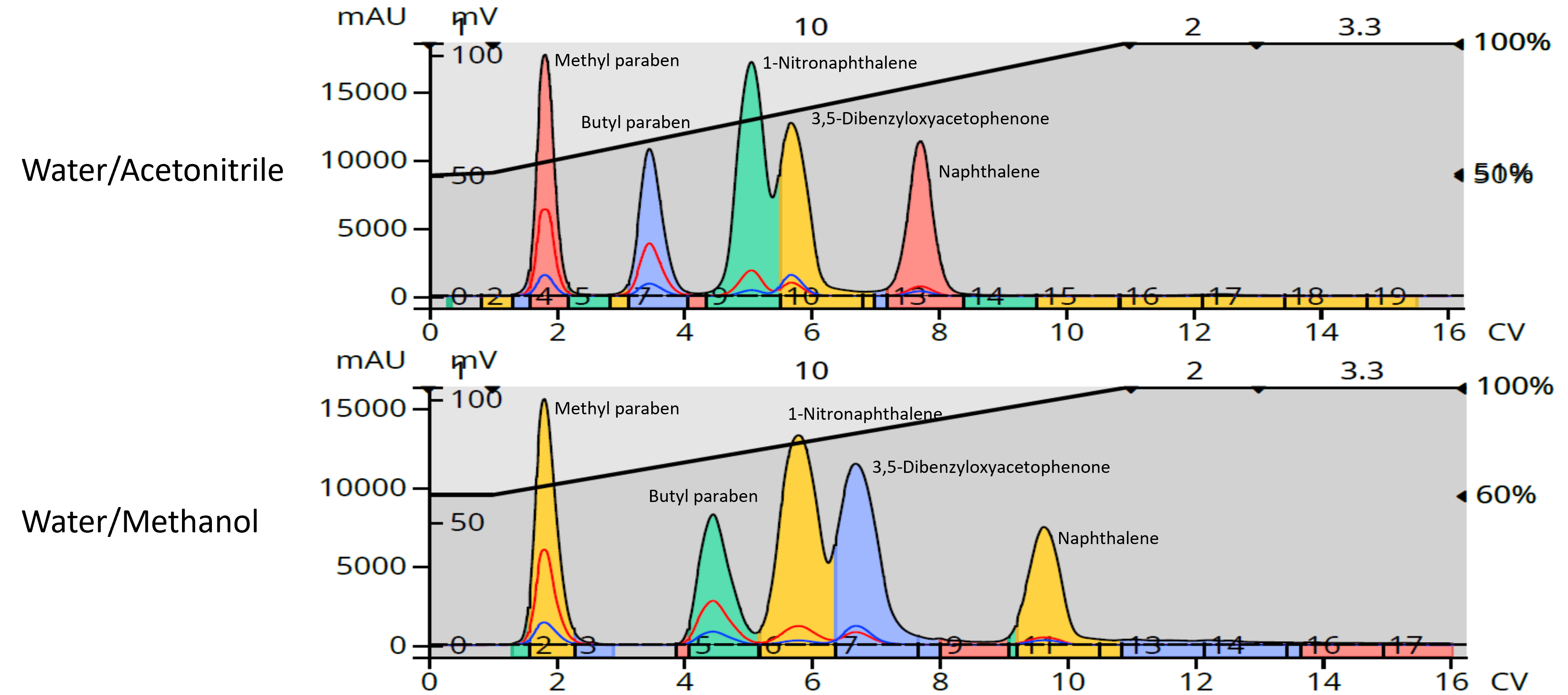
Figure 4. Separation differences are seen between the water/acetonitrile gradient (top) and the water/methanol gradient (bottom).
The first observation is the separation of methyl and butyl paraben. These two compounds are better resolved with the methanol gradient than with acetonitrile even though the methanol percentage is higher than the acetonitrile. Why? It is possible that the parabens are more soluble in acetonitrile, but I don't think so. It is more likely that these two solvents differences in selectivity and protic characteristics, are causing the selectivity differences. The same argument can be made for the last two eluting peaks (3,5-dibenzyloxyacetophenone and naphthalene), which are best separated using the methanol gradient. However, when we look at the separation of butyl paraben and 1-nitronaphthalene (peaks 2 and 3), we see the acetonitrile gradient provides the better separation.
So, regardless of the types of compounds you need to purify, you can evaluate both methanol and acetonitrile for your reversed phase purifications.
Equipment used for this post included…
Biotage® Selekt flash purification system
Biotage® Sfär C18 column, 12-gram
If you are interested in learning more about reversed phase flash, download our white paper - How to Determine Reversed-phase Flash Chromatography Loading Capacity.

