Jan 30, 2023 12:57:54 PM
Five Tips and Tricks for Success in Solid Phase Peptide Synthesis
By Elizabeth Denton

In my role as a peptide application scientist, I have had the pleasure of working with many groups that are venturing into the world of peptides for the first time. Although it seems rather straightforward to experienced synthetic chemists, producing acceptable yield and purity certainly comes with unique challenges in solid phase peptide synthesis .
In this post I would like to present some of the tips and tricks that I have picked up along the way.
I was fortunate when I began my graduate studies to be surrounded by senior graduate students and post docs willing to take the time to help me troubleshoot challenges I encountered as I familiarized myself with solid phase peptide synthesis. Even though there are only three or four relatively straightforward organic reactions that occur throughout the course of synthesizing a peptide, wildly variable results in both crude yield and purity are often observed. These variations are almost always due to the specific peptide sequence itself. I have included in this list some of my most reliable tips and tricks that reliably lead to successful syntheses.
1. Increase the concentration of your amino acid and coupling reagent solution
This should make sense. After all, there are two separate, concentration dependent, bimolecular reactions that must be completed in order to add a single amino acid to a growing peptide chain. By increasing the reagent concentrations you are decreasing the solution volume thereby increasing the probability that the desired molecules will interact appropriately. The effects of amino acid concentration are clear, particularly in the syntheses of longer peptides. I will often increase the concentration of my amino acid and coupling reagent solutions to 0.5 M for most of my peptides.
2. Double couple the incoming amino acid after proline
I have certainly had success single-coupling amino acids in proline-containing peptides, I have also had peptides that fail miserably after a proline residue. The most likely cause is the fact that the reactive nitrogen is a secondary amine and significantly less reactive than the other amino acids. This is most readily confirmed by noting that the pKa of the proline nitrogen is an average of 1 full log unit lower than all other amino acids (10.6 vs average 9.5), with the exception of cysteine.
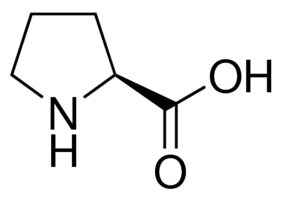
Figure 1: Chemical structure of proline. The reactive nitrogen present as a pyrrolidine, reducing it's reactivity.
3. Double couple when there are multiple identical amino acids in a row
For some peptides that contain only a single amino acid with a bulky sidechain, this makes sense (think Arg8), but you'd be surprised how difficult it can be to synthesize Ala8! Often times these challenges arise due to decreased solubility of the growing peptide or secondary structure formation. However, it can also be challenging to synthesize a peptide that contains only a small number (2-4) of identical amino acids in a linear sequence amongst the remaining peptide sequence. Double coupling the second or third identical amino acid in row can help to reduce the presence of deletion sequences and increase the overall peptide yield and quality.
4. Double couple arginine residues
Arginine can be a challenge to incorporate into any peptide sequence largely due to the bulky side chain and corresponding protecting group. This is particularly prevalent when coupling an arginine to an amino acid with a large sidechain like phenylalanine, tryptophan or tyrosine. An easy way to ensure complete incorporation of the arginine is to double couple, although I have also had some success increasing the allotted coupling time.
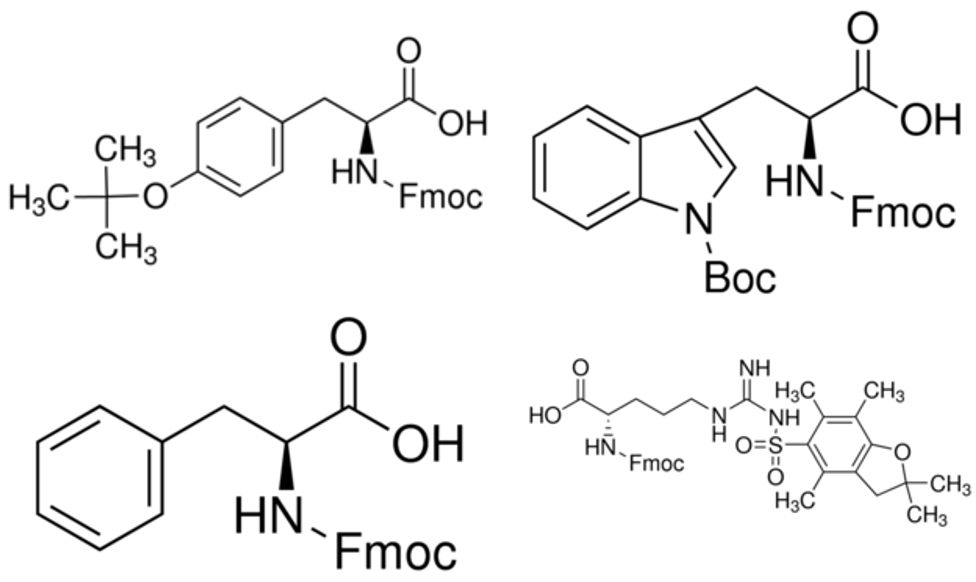
Figure 2: Chemical structure of protected tyrosine (top left), protected tryptophan (top right), protected phenylalanine (bottom left) and protected arginine (bottom right). Fmoc groups have been abbreviated for space considerations.
5. Be aware of aspartic acid residues
Aspartic acid is susceptible to aspartimide rearrangements during the course of peptide synthesis. This side reaction is base catalysed and may occur at any point during every deprotection cycle following incorporation of the aspartic acid residue but has some sequence dependence. The rearrangement introduces a β-amino acid into the otherwise α-amino acid peptide (see mechanism below). Unfortunately, this rearrangement is mass neutral and nearly impossible to separate from the native peptide chromatographically. There have been several strategies published to help limit or eliminate the possibility for this side reaction during synthesis.
/Blogs%20ONLY/aspartimide-3.png?width=443&height=423&name=aspartimide-3.png)
Figure 3: Mechanism for base-catalysed aspartimide rearrangement observed in solid phase peptide synthesis. The iso-Asp by-product is favoured 70% of the time.
To learn more about Biotage tools to improve your peptide synthesis workflow efficiency, click the link below.
Improving Peptide Workflow Efficiency

