Feb 6, 2023 3:14:53 PM
How best to extract reaction products from high boiling solvents
By Bob Bickler

Organic chemistry syntheses often use polar, high boiling point solvents to facilitate high temperature reactions. However, these solvents also can complicate down-stream compound purification, either by evaporation, crystallization, or even flash chromatography.
To isolate the synthetic product from this type of reaction mixture, it must be extracted from the polar reaction solvent. Conventional techniques for this use breakable glass separatory funnels and an immiscible solvent in which the synthetic product hopefully has greater solubility than the reaction solvent. One popular organic extracting solvent is dichloromethane (DCM) because it is denser than most other solvents and solubilizes many organic compounds.
Proper separatory funnel extraction requires many steps [1] which are condensed below.
- Assemble the clean separatory funnel
- Transfer reaction mix into the separatory funnel
- Add DCM and, if necessary water, to create a phase separation
- Gently swirl the DCM + reaction mix to ensure maximum contact between the DCM and the reaction solvent for 60 seconds; avoid emulsion formation
- Invert the separatory funnel, open the stopcock, and allow any gas to escape
- Repeat the swirling and venting 2x
- Insert separatory funnel into holder on a ring stand positioned above a clean beaker or flask
- Open stopcock and slowly collect DCM into the beaker or flask making sure to stop flow at the meniscus or solvent interface
- Add more DCM and repeat steps 4-8 above
- Add magnesium sulfate or sodium sulfate to remove any trace water
- Purify DCM extract or perform solvent exchange prior to purification by reversed-phase
- Clean glass separatory funnel with acetone and/or other solvents, possibly detergents
This process requires ~22 steps (including the repeated steps), several solvents, is time consuming, and hinders workflow.
This post provides insight into a greener, more efficient alternative to glass separatory funnels – recyclable, polypropylene phase separators, Figure 1.

Figure 1. A Biotage® Phase Separator installed on a Biotage® Gravity Rack.
These polypropylene phase separators contain a hydrophobic frit that only allows organic solvents to pass and retains water. Multiple extraction cycles with the same phase separator are possible eliminating the need to “burp” or vent the separatory funnel every extraction. Well-defined aqueous/organic interfaces eliminate the need to stop the extraction just before the solvent interface passes through the stopcock. When finished, the phase separator can be dried and recycled, eliminating the need to clean the device with organic solvents, water, and detergents.
To evaluate the phase separator extraction efficiency, I created my own synthetic product from the reaction of succinic acid plus benzylamine in DMSO using a Biotage® Initiator+ to create N,N’-dibenzylsuccinamide.
To evaluate the phase separator's effectiveness in extracting my product from the DMSO reaction mix, I performed the following steps…
- Transfer 0.5 mL of reaction mixture to a 20-mL scintillation vial
- Add 5-mL water followed by 3-mL DCM to create a clear phase separation
- Shake
- Attach a 25-mL Biotage® Phase Separator into a Biotage® Gravity Rack containing a clean 20-mL scintillation vial (test tubes can also be used)
- Pour the DCM/reaction mix into the phase separator
- Collect the fast-flowing DCM extract (no need to regulate flow rate)
- Add a second 3-mL DCM aliquot into the aqueous solution and collect the DCM
- Evaporate the DCM and re-dissolve in 1:1 methanol/acetone for reversed-phase flash chromatography
- Purify with flash chromatography
- Air dry and recycle the phase separator
Ten steps, the longest of which is the 10-minute flash purification. Nothing to clean, no need to monitor separatory funnel flow rate and solvent interface, and no expensive, breakable glassware. See video below...
I evaluated the extraction effectiveness using reversed-phase flash chromatography. The chromatography showed successful extraction, and separation, of several compounds from the DMSO reaction solvent, Figure 2.
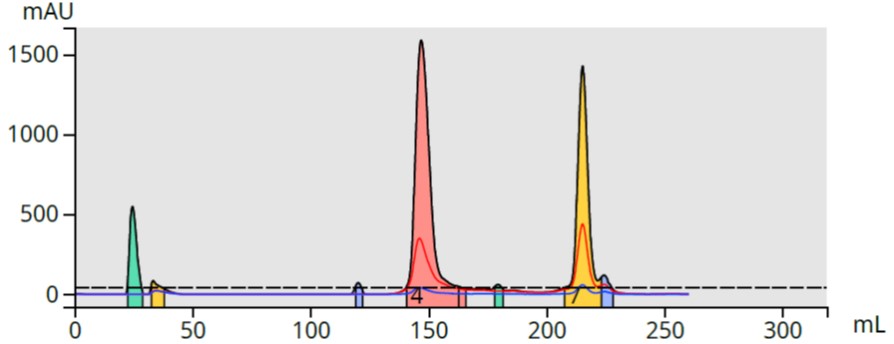
Figure 2. DCM extract flash chromatography shows the presence of two major peaks and a few minor by-products.
To determine the extraction efficiency, the aqueous portion was also subjected to flash chromatography using the same method and showed nearly complete extraction of the reaction products, Figure 3.
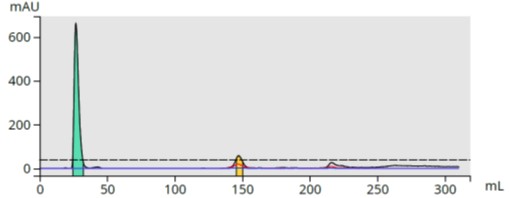
Figure 3. Upper aqueous layer reversed-phase flash chromatography shows minimal amounts of reaction by-products.
Because the same recyclable extraction vessel allows multiple extractions of the same reaction mixture, which take only seconds, these phase separators are used by many pharmaceutical companies for synthetic research, increasing their productivity while reducing research costs.
For more information on Biotage workup products, click the link below.
[1] https://chem.libretexts.org/Bookshelves/Organic_Chemistry/Book%3A_Organic_Chemistry_Lab_Techniques_

