Dec 5, 2023 3:00:00 PM
How can I remove color from my reaction product?
By Bob Bickler

Have you ever had a purified reaction product, which should be white, retain color after purification? If so, what did you do to remove it?
I came across this situation recently when I was synthesizing 2,4,6-trichloroaniline [1] using a microwave reactor, Figure 1.
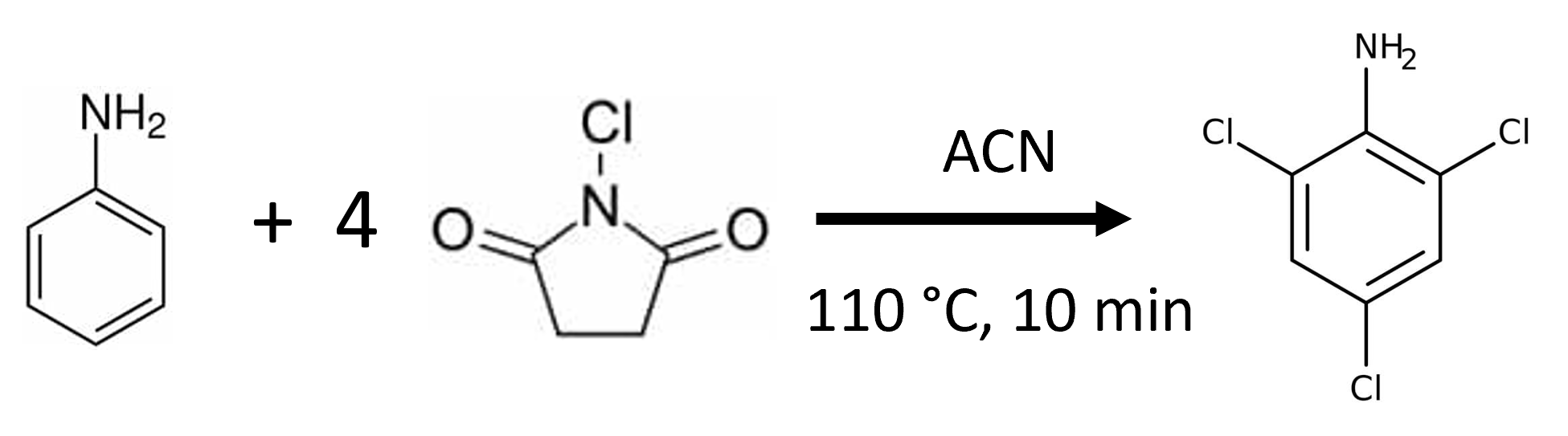
Figure 1. Microwave reaction of aniline with N-chlorosuccinimide.
The reaction yielded a rust-colored crude mixture. TLC indicated the product and several by-products were created. Normal phase flash chromatography using heptane and methyl-t-butyl ether (MTBE) looked promising but with the reaction performed in acetonitrile, which is immiscible with heptane, I needed to extract the product out of the reaction solvent and redissolve in a heptane compatible solvent such as dichloromethane (DCM). Extraction would also remove some polar by-products and excess N-chlorosuccinimide.
After extraction with DCM, I added silica and evaporated the solvent to create a dry load (~300 mg). During purification of the extract, a very tight, rust-colored band moved through the silica column, Figure 2.
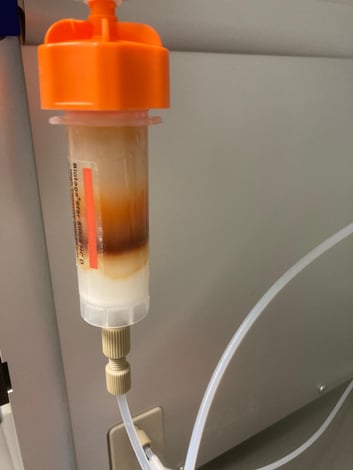
Figure 2. Normal phase flash purification of the reaction mixture DCM extract (300 mg) showed the product eluting in a very tight, highly colored band.
This band generated a good chromatogram with few separated by-products, Figure 3.
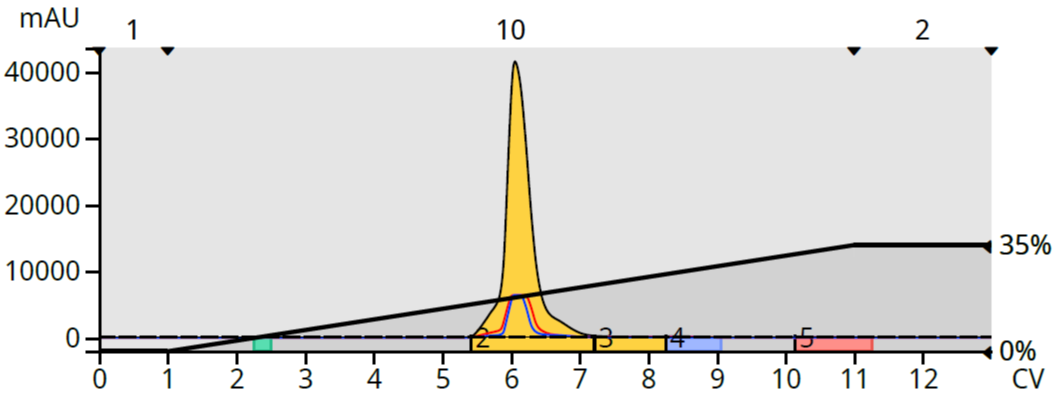
Figure 3. Normal phase flash purification of 300 mg of synthetic 2,4,6-trichloroaniline using a 0-35% MTBE in heptane gradient.
From the 300 mg loaded, 183 mg of red crystals were recovered, indicating normal phase removed a large number of impurities.
So, the product is pure, right? Well, not quite. According to the Davis article, the product should be “soft, colorless needles”, not red crystals. As stated in his article, to remove the red color from the product, he used both activated carbon and silica, without supplying much detail about the color removal protocols.
I could have tried repurification with silica and less polar solvents like heptane with DCM but decided on a greener solution using reversed phase flash. After minimal method development I found a 60-100% methanol gradient to be promising, Figure 4.
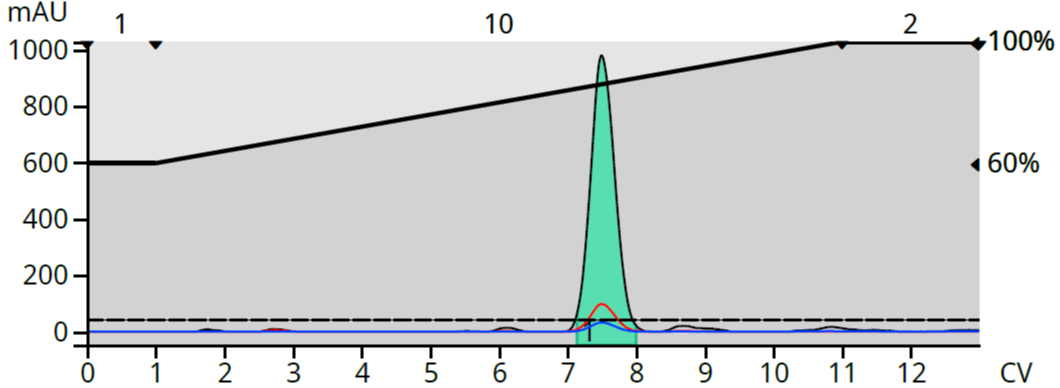
Figure 4. Reversed phase scouting run of normal phase purified product.
I dried the silica-purified red product (183 mg) onto C18 media, packed it into a dry load vessel, and repurified on a 12-gram C18 column. The chromatogram showed a good separation with the product separated from several impurities, Figure 5.
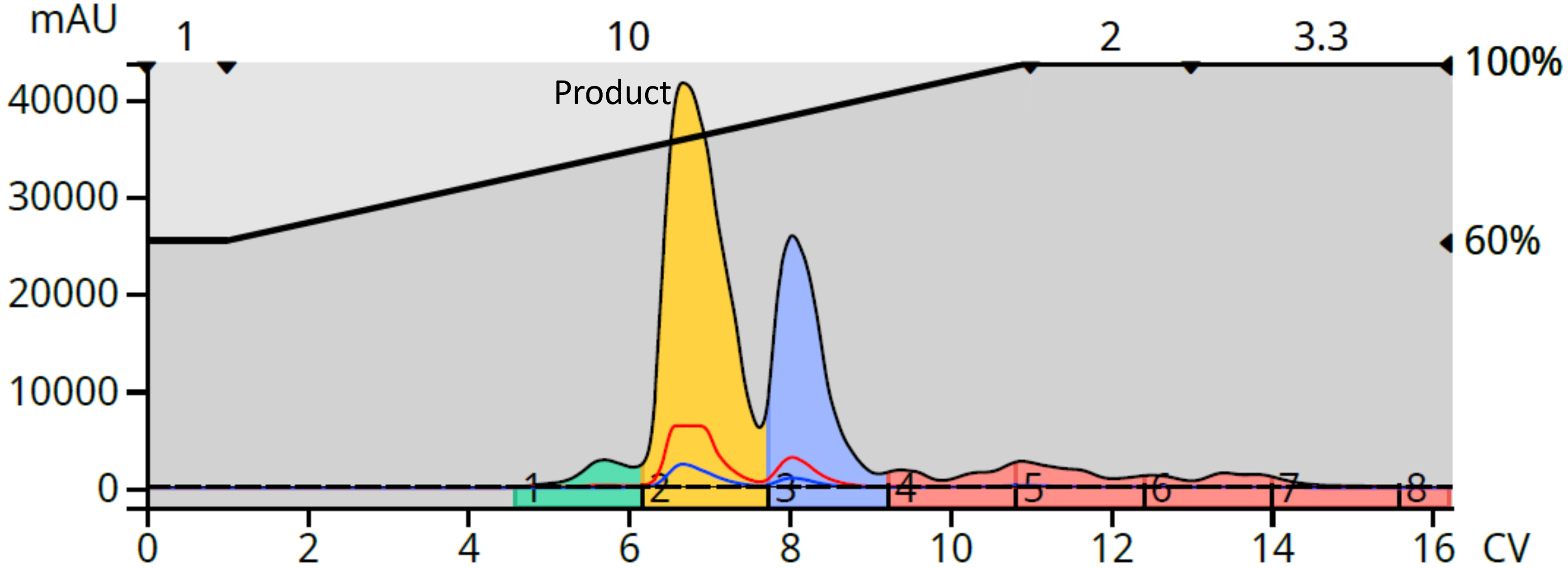
Figure 5. Reversed-phase flash purification of the normal phase product fraction.
The collected fractions indicated the color was removed from the product which eluted in fraction 2 while the colored fractions eluted later, beginning with fraction 4, Figure 6.

Figure 6. Reversed phase purification fractions. The product fraction was clear and eluted at fraction 2 while the colored impurities eluted later in fractions 4-7.
The product in fraction 2 was evaporated yielding 58 mg of soft, colorless needles, Figure 6.

Figure 6. 2,4,6-Trichloroaniline final product.
For this work, I used the following equipment:
- Biotage® Initiator+ microwave reactor
- Biotage® V-10 Touch solvent evaporator
- Biotage® Selekt Enkel flash chromatography system
- Biotage® Sfär HC (10 gram) and C18 (12 gram) flash columns
- Biotage DLV (dry load vessel) with Biotage® KP-Sil and KP-C18-HS
So, if you have color contamination in your product and have previously only purified it using silica, consider repurification by reversed-phase flash chromatography. It’s simple, easy, and greener than other techniques.
If you are interested in learning more about reversed phase flash chromatography, download our whitepaper How to Determine Reversed Phase Flash Chromatography Loading Capacity.
[1] Matthew C. Davis (2009) Chlorination of Aniline and Methyl Carbanilate by N-Chlorosuccinimide and Synthesis of 1,3,5-Trichlorobenzene, Synthetic Communications®, 39:6, 1100-1108, DOI: 10.1080/00397910802499542

