Nov 21, 2023 3:00:00 PM
How does an acid pH modifier impact flash chromatography?
By Bob Bickler

With the increased use of reversed phase flash chromatography for reaction mixture purification comes a lot of questions since many chemists are not as familiar with this technique as they are with normal phase or silica column chromatography. One of the questions chemists ask is about the use of pH modifiers, specifically acids, with their purification.
In the HPLC world where reversed phase dominates, use of an acid (formic, acetic, or trifluoroacetic), typically at a 0.1% level, is commonplace. The rationale is that if a sample component is ionizable in solution, its chromatography retention and peak shape can be impacted, and its quantitation challenged.
Ionized/ionizable compounds are in equilibrium between their charged and neutral states when in solution. These molecules, when chromatographed in a neutral solvent system, can show as a fronting peak (think ski slope) because the ionized portion is more water soluble than the neutral state and thus elutes faster. Adding a pH modifier, whether an acid or base, can force the equilibrium either fully into the neutral state or into an ionized state. When fully ionized, the compound will elute early; when neutral, the compound is more hydrophobic and its retention on the column enhanced.
For example, let’s look at the purification of synthetic N-(1-phenylethyl)succinimide created through the microwave reaction of succinic anhydride and α-methylbenzylamine, Figure 1.
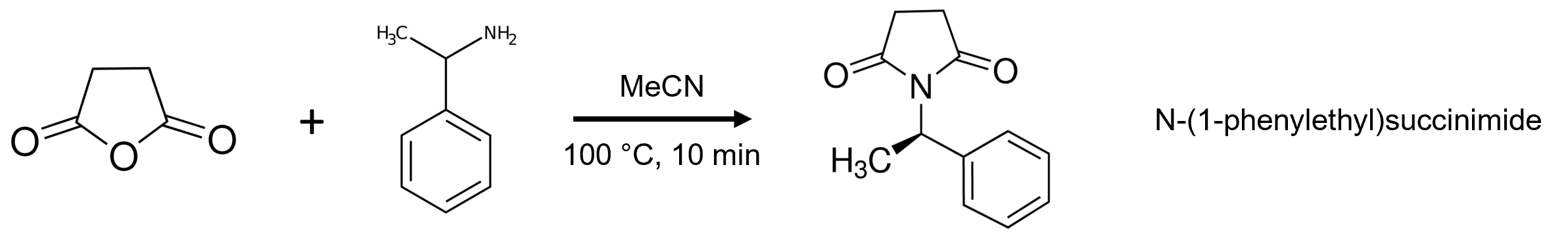
Figure 1. Succinic anhydride reaction with α-methylbenzyl amine using a Biotage® Initiator+ microwave reactor
When purified using neutral solvents, the product fronts badly indicating it is in equilibrium between an ionized and neutral state, Figure 2.
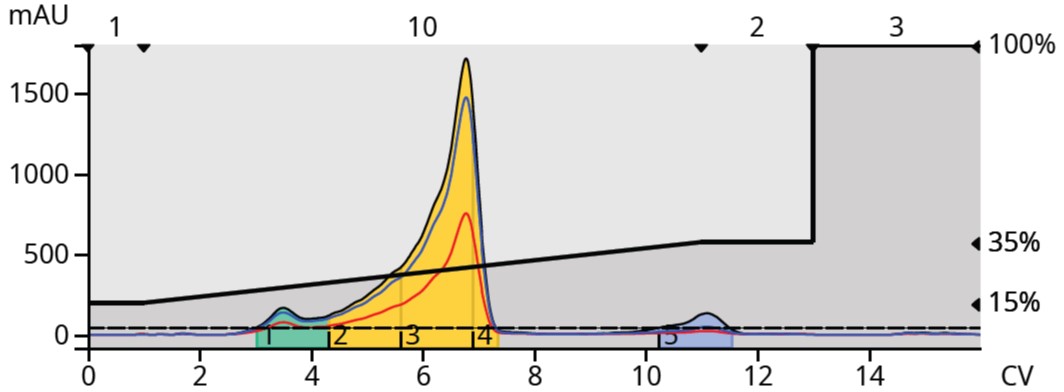
Figure 2. Reaction mixture reversed phase flash chromatography using neutral water and acetonitrile showed a fronting product peak.
The issue with the product eluting as a non-gaussian, fronting peak is that it is a broad and collected over several column volumes (CV). This means the collected product is diluted in more solvent than is typical or desired which increases the volume and time to evaporate the fraction solvent and increases the potential for lower purity and recovery. In fact, with this reaction mixture there is an earlier eluting byproduct which is contaminating the early product fractions. Yes, those fractions could be discarded but that reduces the recovered product yield, not necessarily a desirable solution.
By introducing an acid, in this case 0.2% glacial acetic, to both water and acetonitrile, the ionizable reaction mixture components protonated . The protonated product’s elution band reduced (sharpened) which increased its concentration and separation from the contaminating byproduct, Figure 3. The byproduct also protonated causing it to elute earlier than with the neutral mobile phase. The result is a vastly improved separation with the product eluting in a more concentrated band with better resolution from the byproducts.
Figure 3. Reaction mixture purification using an acidified solvent system sharped the product peak and decreased retention of an interfering byproduct.
Chemists do, however, need to be careful when using pH modified mobile phases because of potential compound retention shifting with pH change. In the previous example, ionized compound shifting was helpful, but protonated compounds don’t always move in beneficial directions. When pH is changed there is a chance that eluting compounds move closer to each other or even coelute.
We can see this occur with the synthetic product 2-amino-N-(1-phenylethyl)benzamide created by the microwave reaction of isatoic anhydride and α-methylbenzyl amine, Figure 4.
Figure 4. Microwave reaction of isatoic anhydride with α-methylbenzylamine
When purified using a neutral water/methanol gradient, we can see some polar by-products eluting prior to the product as well as a strongly retained by-product eluting as a broad peak behind the product between 9.5 and 14 CV, Figure 5.
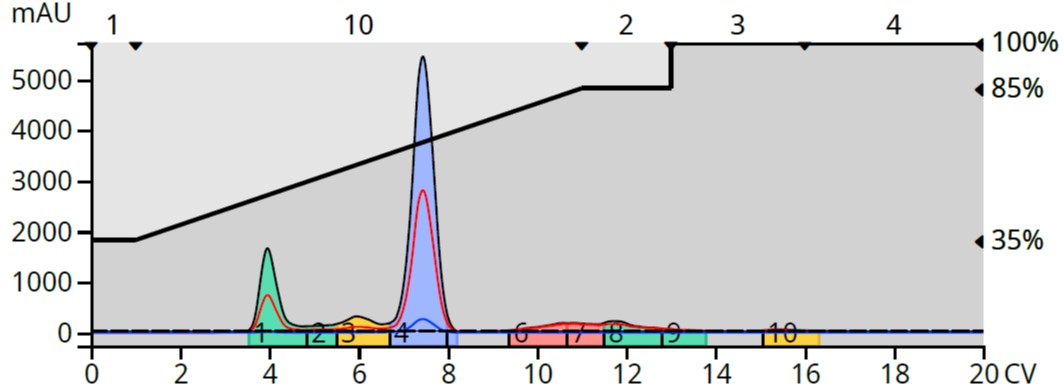
Figure 5. pH neutral reversed phase flash chromatography of the reaction mixture
With the addition of 0.2% glacial acetic acid to both elution solvents, there is a dramatic shift in eluting compound retention. A by-product eluting at 4 CV under neutral pH, ionized and eluted at 1.5 CV under acidic conditions. The trailing hydrophobic by-product also ionized and moved closer to the product, Figure 6.
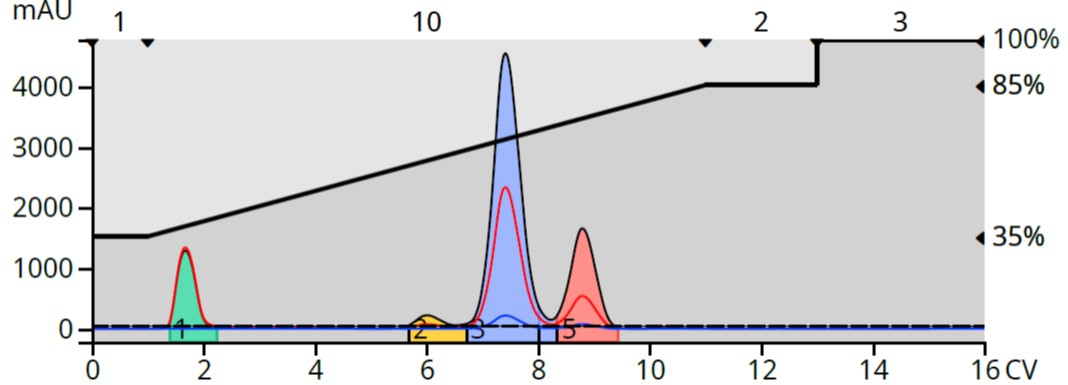
Figure 6. Reaction mixture purification with an acidified water/methanol gradient shifted retention of two by-products.
With the reduced resolution between the product and the trailing by-product, the loading capacity for this reaction purification decreases meaning larger columns (and more solvent) are needed to sufficiently purify the reaction product.
So, while mobile phase pH adjustments can help improve your purification, be aware that elution volumes and selectivity can change, potentially reducing your purification efficiency.
Equipment used to create this data included:
- Biotage® Initiator+ microwave reactor
- Biotage® Selekt Enkel flash chromatography system
- Biotage® Sfär C18 column, 12-gram
Interested in learning more about flash chromatography? Download our white paper Inspiring Productivity with Modern Flash Chromatography.
Published: Nov 21, 2023 3:00:00 PM

