Oct 25, 2023 2:00:00 PM
How much methanol can I use with my silica flash column?
By Bob Bickler

Silica dissolution in methanol is a concern many chemists have. Over my career I have heard that complaint probably hundreds of times which invariably leads to the question - how much methanol can I use with may silica column?
Well, if you research this topic, you will find that silica is not dissolved by methanol [1]. It will, however, start dissolving in methanol if the solvent’s alkalinity increases, say with the addition of ammonia. So, DCM/MeOH/ammonia mobile phases can cause silica dissolution. The higher the pH, the faster the rate of dissolution.
But if methanol itself does not dissolve silica, then why do chemists using neutral DCM/MeOH mobile phases often find silica particles in their fractions? The reason is silica quality and its manufacturing processes.
There are two silica types used in chromatography, granular and spherical. Traditional flash silica has a granular or irregular shape, Figure 1. This shape is created by grinding larger particle size, synthetic, spheroidal silica into smaller particles which are then sieved to the appropriate particle size for flash (40-63 µm).
 Figure 1. Granular (irregular) silica.
Figure 1. Granular (irregular) silica.
This grinding process exerts tremendous pressure on the particles increasing their friability, which is exacerbated by passing dense solvents, like DCM, and polar solvents, like methanol, through them. So, when using granular silica in a flash column, DCM/MeOH elution gradients can cause weak spots in the silica to breakdown into fines (very small particles) which can pass through the column’s bottom frit or filter and into product fractions.
Spherical silica, however, is manufactured to be the desired particle size and, therefore, does not require the grinding process, Figure 2.
 Figure 2. Spherical silica.
Figure 2. Spherical silica.
Because these particles never experience the grinding pressures seen by the granular silica, they tend to be far more tolerant of DCM/MeOH solvent gradients, even in 100% methanol.
Tests performed at Biotage with a commercially available granular (brands X and Y) and Biotage spherical silica (lot 231105, columns 0826 and 0916), 10 - 12 g sizes, showed that spherical silica did not shed silica particles when subjected to a DCM/MeOH gradient (0-100% MeOH in DCM), Figure 3. Biotage only manufactures columns packed with spherical silica.
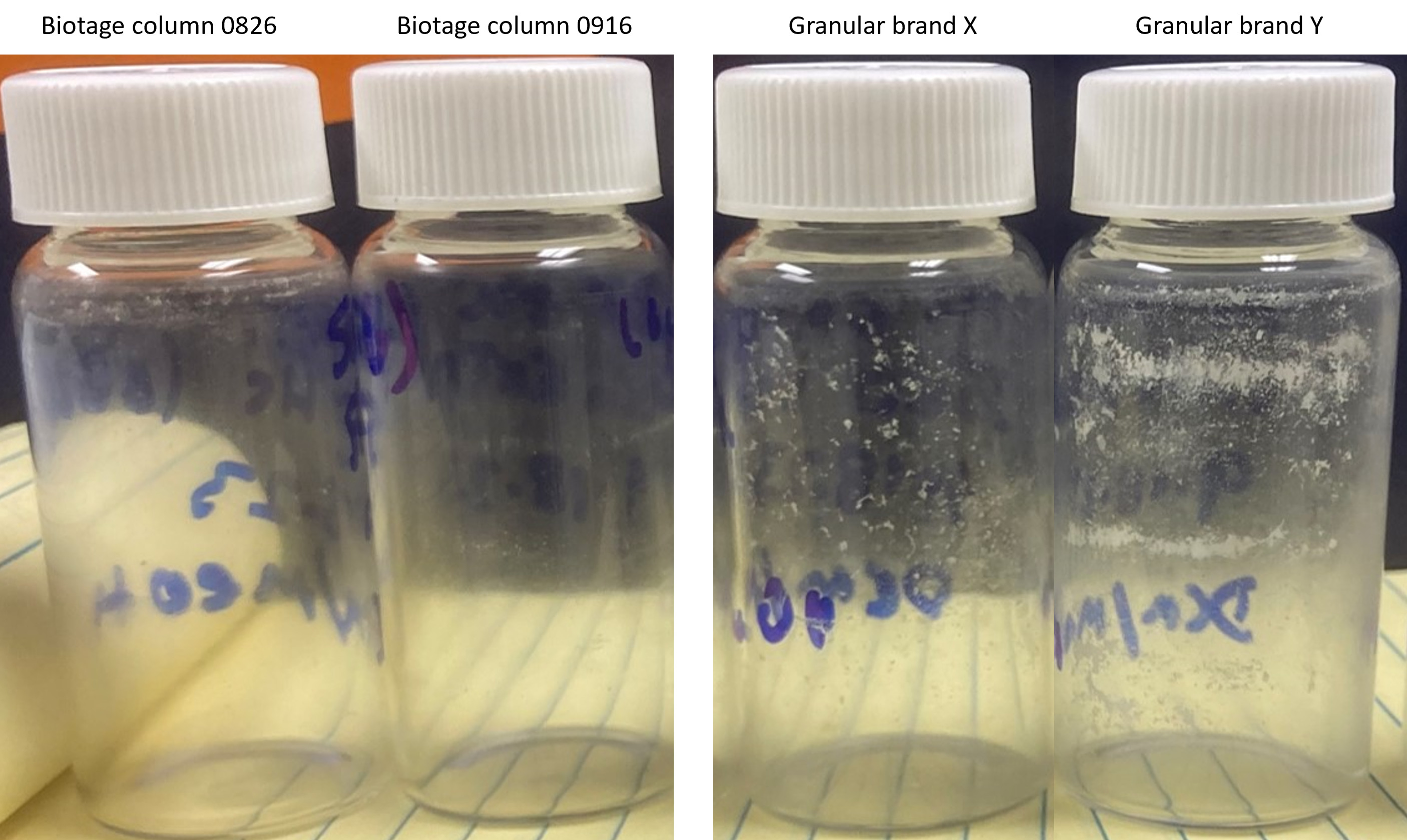 Figure 3. DCM-MeOH silica column extracts. Left - Biotage spherical silica extract. Right - brand X, and brand Y granular silica extracts. Only the granular silica columns shed particles.
Figure 3. DCM-MeOH silica column extracts. Left - Biotage spherical silica extract. Right - brand X, and brand Y granular silica extracts. Only the granular silica columns shed particles.
The tests showed that both granular silica columns shed silica particles with brand Y shedding more than brand X. The results also showed the Biotage spherical silica columns did not shed any silica particulates.
So, if you need to use DCM/MeOH elution gradients for reaction mixture purification, use spherical silica columns to minimize silica contamination of your product.
Interested in learning more about flash chromatography? Download our updated whitepaper – Successful Flash Chromatography.
[1] Solubility of silica (silicon dioxide) in methanol - Chromatography Forum (chromforum.org)

