Jan 9, 2024 3:00:00 PM
How short can I make my flash chromatography method?
By Bob Bickler

We all know the adage “Time is Money”. It is particularly true in pharma where the timeline to get a pharmaceutical product to market can take 10+ years and cost billions of dollars. With this as the backdrop, anything that can be done to speed the drug discovery to commercialization process helps reduce costs.
I have mentioned in past posts several techniques that can be incorporated to incrementally achieve this goal including step gradients. What I want to talk about is another approach which may be easier and equally effective – shorter linear gradients.
Most flash chromatography methods are linear gradients spanning 10 or more column volumes (CV). This gradient length often is more than what is needed for a particular purification and thus wastes time and solvent.
A very good technique to reduce run time and solvent is the step gradient as mentioned above. While I am a fan of step gradients, they can be challenging to develop with reversed phase flash purification, an increasingly popular purification technique.
Thin-layer chromatography (TLC) is a great method development tool for both linear and step gradients for normal phase (silica column) chromatography. However, its usefulness for reversed phase method development is limited due to its hydrophobic surface wettability issues with highly aqueous mobile phases.
For many chemists, reversed phase flash chromatography methods are either transferred from HPLC systems or created through trial and error. These purification methods can be longer than required, especially those linear or isocratic methods transferred from HPLC systems that are time-based rather than CV-based.
Depending on your purification goals (purity/throughput) and the resolution between your target compound and its closest eluting byproducts, a shortened linear gradient may meet your needs. Just reducing the method’s gradient length, however, is not the best approach. If you maintain the gradient starting and ending %B solvent and reduce the gradient length, the gradient slope will change and that can negatively affect the results by reducing amount of separation, or resolution, between eluting compounds, Figure 1.
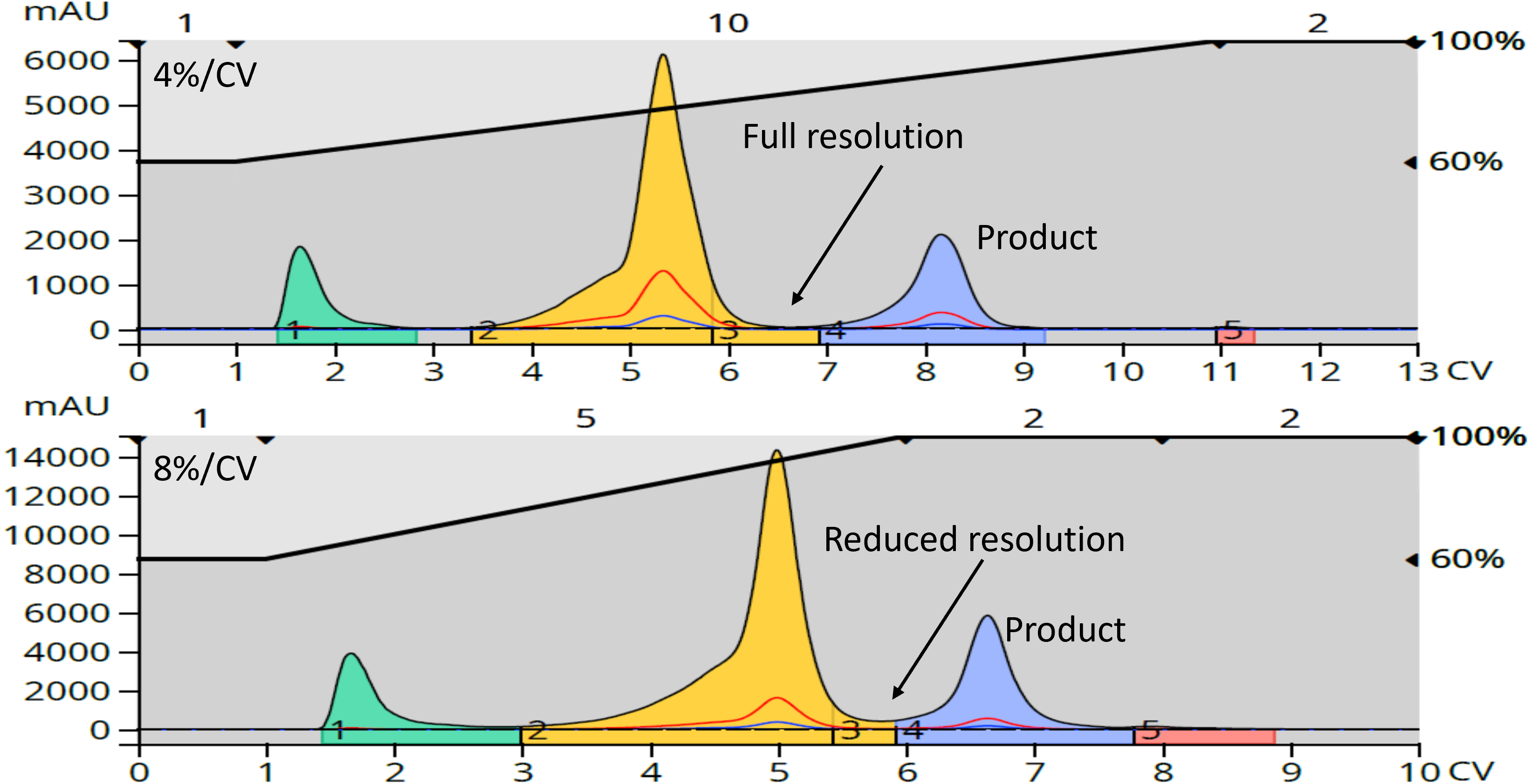 Figure 1. Impact of a shortened linear gradient without accounting for slope is reduced separation. Top - 4% B/CV. Bottom - 8% B/CV.
Figure 1. Impact of a shortened linear gradient without accounting for slope is reduced separation. Top - 4% B/CV. Bottom - 8% B/CV.
To minimize the resolution loss, it is much better to maintain a constant gradient slope (%B/CV) by adjusting either the starting %B, ending %B, or both when shortening the purification method. The gradient length can then be adjusted to best suit your needs, Figure 2.
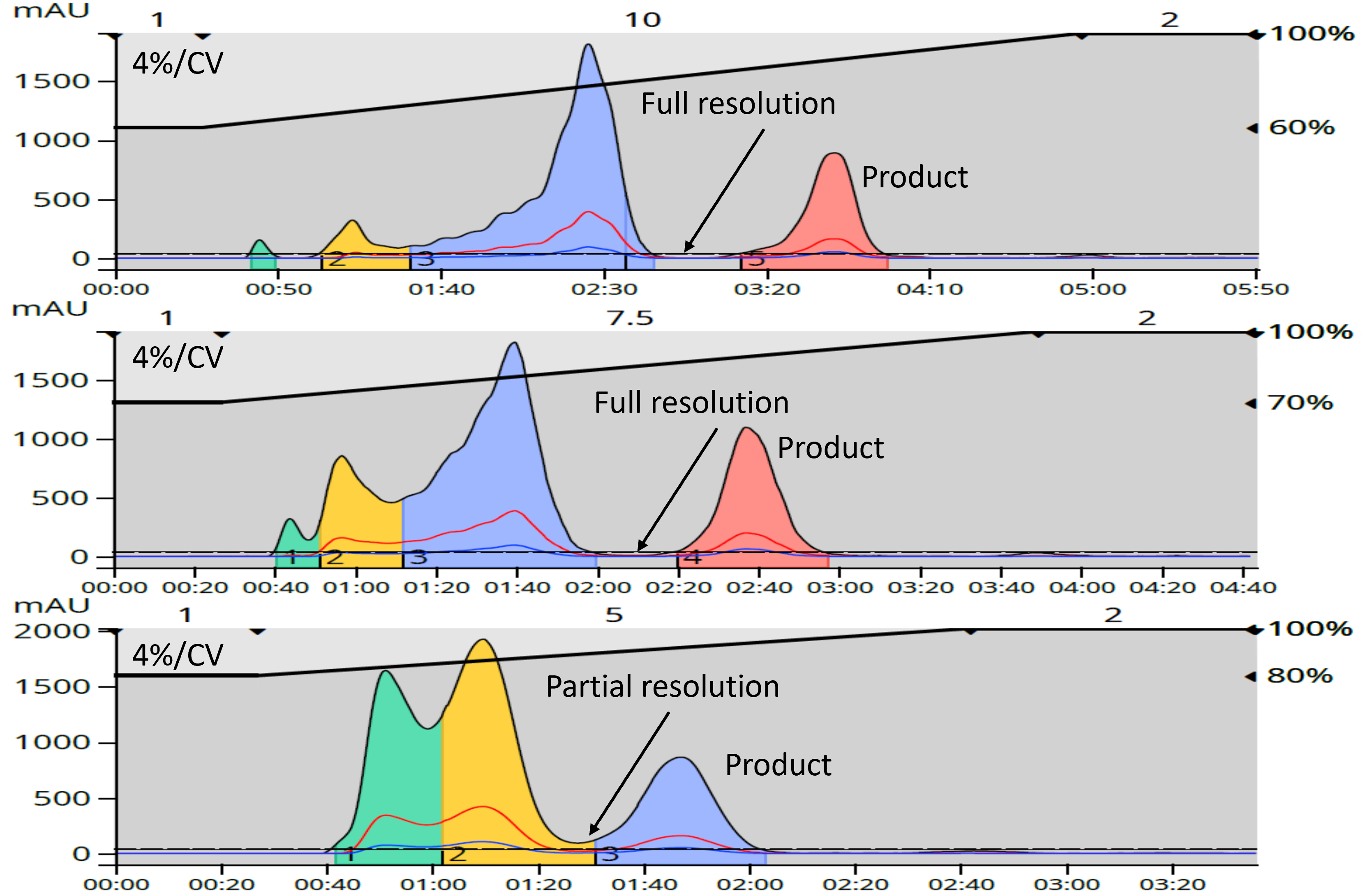 Figure 2. Impact of gradient length at constant slope.
Figure 2. Impact of gradient length at constant slope.
Remember, the separation of compounds is a balance between compound residence time on the column media and its solubility in the mobile phase so shortened gradients can, even with constant gradient slope, degrade the separation.
From the data above in Figure 2, a complete separation was still achieved with a 7.5 CV gradient by maintaining the original method’s 4%/CV slope. Depending on your product purity goals, even the 5 CV gradient may be a sufficiently long run.
The benefits of these shorter gradients include both reduced purification time and solvent use, Table 1.
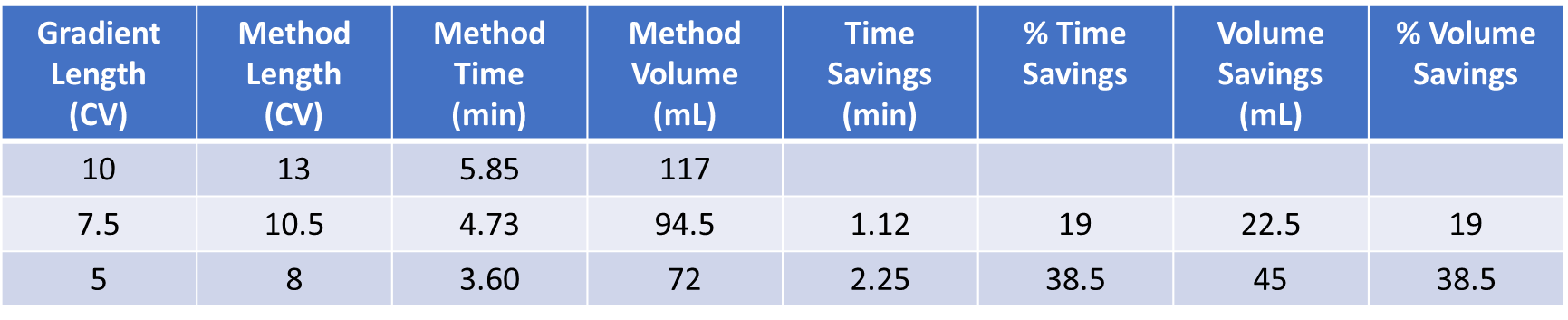 Table 1. Time and solvent savings possible with short linear gradients at a constant slope.
Table 1. Time and solvent savings possible with short linear gradients at a constant slope.
So, if you need to reduce purification time, and/or solvent, and your initial separation provides sufficient resolution, consider reducing the gradient length while maintaining slope.
All of the above examples used a 6-gram Biotage® Sfär C18 column operated at 20 mL/min on a Biotage® Selekt Enkel system.
For more information on flash chromatography, download our whitepaper - Some Tips for Achieving Greener, Safer Flash Chromatography.

