Feb 7, 2023 10:16:17 AM
How to handle peptides that contain methionine
By Elizabeth Denton

The diversity of amino acid side chain functionalities, coupled with secondary structure, gives peptides and proteins their unique properties and activities. However, when it comes to chemically synthesizing peptides or even small proteins, the side chain functionalities can do more harm than good.
In today's post, I'll discuss synthesizing peptides that contain methionine and how to overcome a potentially serious side reaction involving this amino acid.
Methionine and cysteine are the only two amino acids within the naturally occurring twenty that contain a sulfur atom in the side chain which provides for unique reactivity profiles. Cysteine is well known for its ability to form disulfide bonds - covalently stabilizing secondary structures. From a synthetic standpoint though, the Cys thiol is protected (often with a trityl protecting group) reducing its susceptibility for undesired side reactions during synthesis.
Methionine however, has most notably become a marker for oxidative stress, as the thioether side chain is highly susceptible to oxidation by a variety of reactive oxygen species. The methionine thioether has been found to be non-reactive under standard Fmoc-based solid phase peptide synthesis and hence is not protected. Unfortunately though, the thioether is still sensitive to the environment during both synthesis and cleavage from the resin support.
I was doing some synthesis optimization for the amyloid-beta peptide (residues 1-42) and decided to do an experiment. I have done little work with peptides containing methionine in the past and was curious to what extent Met oxidation can occur. Not only is amyloid-beta a notoriously difficult peptide to synthesize, it also contains a methionine residue eight residues from the C-terminus. I chose not to bathe the amino acid solutions and reactor vial in an inert atmosphere during synthesis and cleaved the peptide from the resin using a modified Reagent B cocktail (95:2.5:2.5 TFA:water:TIS). Under these conditions I observed significant formation of methionine sulfoxide (indicated by mass increase of 16 Da relative to the parent molecular weight) in my own syntheses, Figure 1.
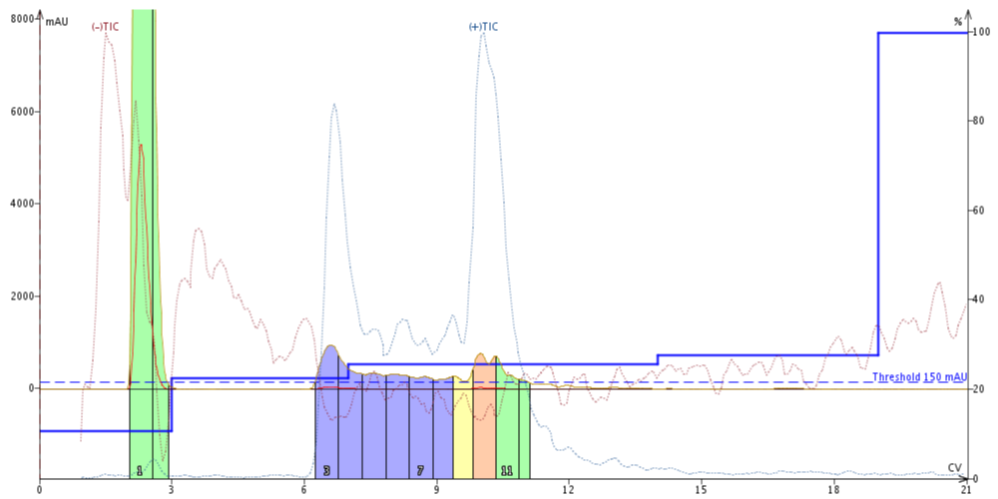
Figure 1. Crude sample analysis of Amyloid-beta (1-42). The solution contains approximately 50:50 desired product:oxidized peptide when no preventative measures are taken.
There are some well established strategies to either reduce or eliminate the Met oxidation, and full disclosure, I avoided all of them for this synthesis. So how do you deal with this? Biology has clearly found a way to reverse this reaction, is there a chemical process that can also be applied?
Given the fact that Met oxidation occurs primarily during acidic cleavage from the resin, an alternative cleavage cocktail was developed by Huang and Rabenstein in the late 90s. They found that the addition of dimethylsulfide and ammonium iodide significantly reduced or eliminated the presence of methionine sulfoxide without significantly compromising yield.
A similar set of reagents has been similarly used to reduce methionine sulfoxide in solution if necessary. An option like this is particularly important as synthesis of peptide fragments are ligated via a native chemical ligation mechanism while still preserving the C-terminal thioester.
Although there are a couple additional chemistry steps involved, it is somewhat of a relief to know that there are some options to reversing this side reaction that doesn't involve resynthesizing the entire peptide.
There are some cases where oxidation is a good thing - like the formation of disulfide bonds. Click below to learn more about automating disulfide bond formation on resin.

