Jan 23, 2023 10:41:52 AM
Setting the Right Flow Rate for Flash Column Chromatography
By Bob Bickler

How does flow rate impact my flash column chromatography separation? This is the kind of question I frequently get. After all, we all know that flow rates that are too high or too low can result in bad prep HPLC chromatography. Well, this is not necessarily true in flash chromatography.
In chromatography there are three inter-related variables which impact your separation and are represented on the chromatographer’s triangle.
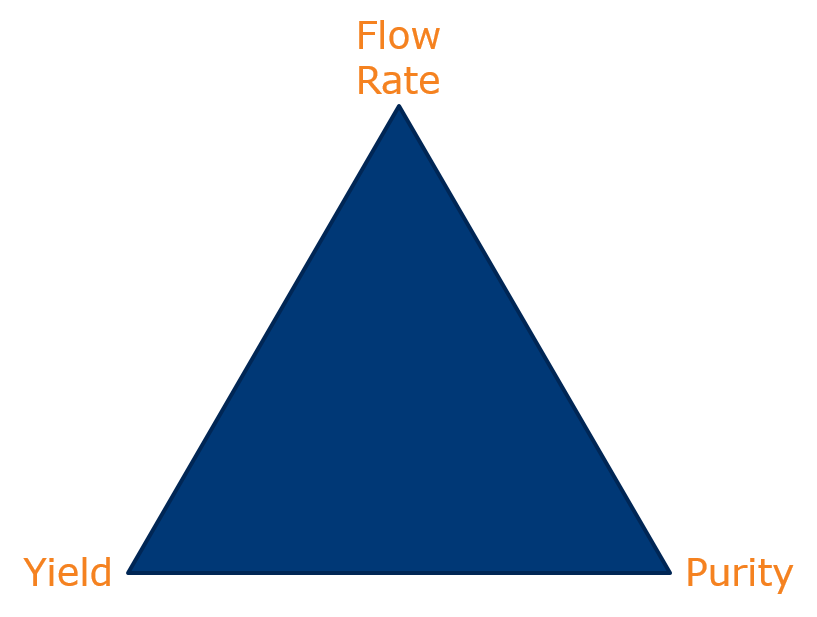 In practice, only two of these variables can be optimized. Increasing flow rate decreases either load (if you need to maintain a purity level) or purity (if you need to maintain a throughput) because of increased sample dispersion and band-broadening. If you increase load, you may need to change the flow rate or purity will suffer. You get the idea.
In practice, only two of these variables can be optimized. Increasing flow rate decreases either load (if you need to maintain a purity level) or purity (if you need to maintain a throughput) because of increased sample dispersion and band-broadening. If you increase load, you may need to change the flow rate or purity will suffer. You get the idea.
In this post, I will discuss the impact that flow rate has on flash chromatographic performance.
In previous posts I have talked about flow rate and linear velocity and how it is important to maintain linear velocity when scaling up a purification. Until now, though, I have not discussed the impact flow rate has on linear velocity and, likewise, load and purity.
Flash column chromatography is a vital part of the organic or natural product chemist’s tool box. An organic or medicinal chemist may only need a target intermediate compound at 80% purity, while a natural product chemist may need better than 95% purity.
Students of chromatography know about the van Deemter curve and how it models the correlation between column efficiency (plate count) and flow linear velocity. For every particle size media used for a stationary phase there is a unique, optimal linear velocity. Generally, smaller media particles have higher optimal flow rates. Additionally, the optimal flow rate range is increased as well. When the flow rate is optimized so is the loading and fraction purity due to a better separation between compounds in the mix. However, the optimal flow rate for a column typically translates to a relatively low flow rate thus a longer purification time.
Trade-off decision time. Ask yourself, what is more important – fast purification with high yield or a high purity product with high yield? So, if you want to purify your sample as fast as possible with maximum purity and yield (the ideal situation) you will need to increase column size because your purification will be load limited (but that increases purification costs). But if purity is the priority, then use a flow rate that gives the optimal linear velocity for any specific flash column.
So what is “optimal” when it comes to flow rates on columns packed with different particle size media? Well, there is published data [1], primarily for analytical HPLC columns, which shows the optimal linear velocity is about ~0.8 mm/sec for a 10 micron media and ~1.8 mm/sec for a 5 micron particle. Bigger particles such as those used in flash and column chromatography demand even slower linear velocities to be optimal. However, because of these slower than ideal flow rates flash column manufacturers have moved to smaller, more efficient 20-25 µm flash media in recent years. After all, flash chromatography is supposed to be fast, right?
So, what do these linear velocities translate to in terms of flow rate? Well, if we assume a 50 µm particle has an optimal velocity of 0.1 mm/sec and a 25 micron has an optimal velocity of 0.3 mm/sec then your flow rates are ridiculously slow, much too impractical for routine use. How slow? See the Table below.
|
Column size (g) |
50 µm media |
25 µm media |
|
10 |
2 mL/min |
6 mL/min |
|
25 |
3 mL/min |
12 mL/min |
|
50 |
6 mL/min |
21 mL/min |
|
100 |
6 mL/min |
21 mL/min |
|
340 |
20 mL/min |
70 mL/min |
Ouch, really low flow rates. So then, what is a reasonable and practical linear velocity for flash-grade media that will provide sufficient loading capacity, separation performance (purity), and purification time?
Well, most flash column manufacturers have developed and published flow rate ranges they believe are practical. For flash chromatography, as with HPLC, the optimal flow rate is particle size dependent.
To study the impact of flow rate on separation performance I performed a test where I separated a mix of two compounds, ethyl and propyl paraben, by reversed-phase flash chromatography at four flow rates from 20 to 50 mL/min in 10 mL/min increments.
I used two different cartridges, a 12 gram Biotage® SNAP KP-C18-HS with a nominal 50 µm particle media and a 12 gram Biotage® SNAP Ultra C18 packed with a 25 µm media. To eliminate gradient effects, I performed the separation using an isocratic 50:50 methanol/water mobile phase and maintained the same sample load, 50 mg per run.
The results obtained showed a pronounced separation (resolution) decline with increasing flow rate with the larger 50 µm particle media, Figure 1. The smaller 25 µm particle media showed separation stability from 20 through 40 mL/min with only a slight decline at 50 mL/min, Figure 2.
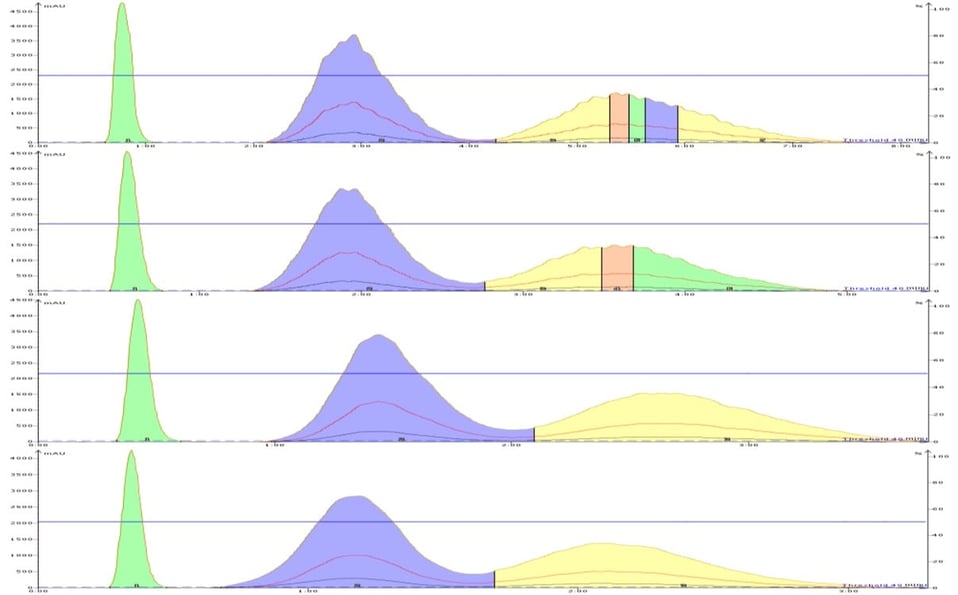 Figure 1. Separations obtained of a 2-component paraben mix by a reversed-phase, 50 micron, 12 gram, Biotage KP-C18-HS column at 20, 30, 40, and 50 mL/min (top to bottom) show a maximum separation at 20 mL/min as indicated by its nearly baseline separation between peaks 2 and 3.
Figure 1. Separations obtained of a 2-component paraben mix by a reversed-phase, 50 micron, 12 gram, Biotage KP-C18-HS column at 20, 30, 40, and 50 mL/min (top to bottom) show a maximum separation at 20 mL/min as indicated by its nearly baseline separation between peaks 2 and 3.
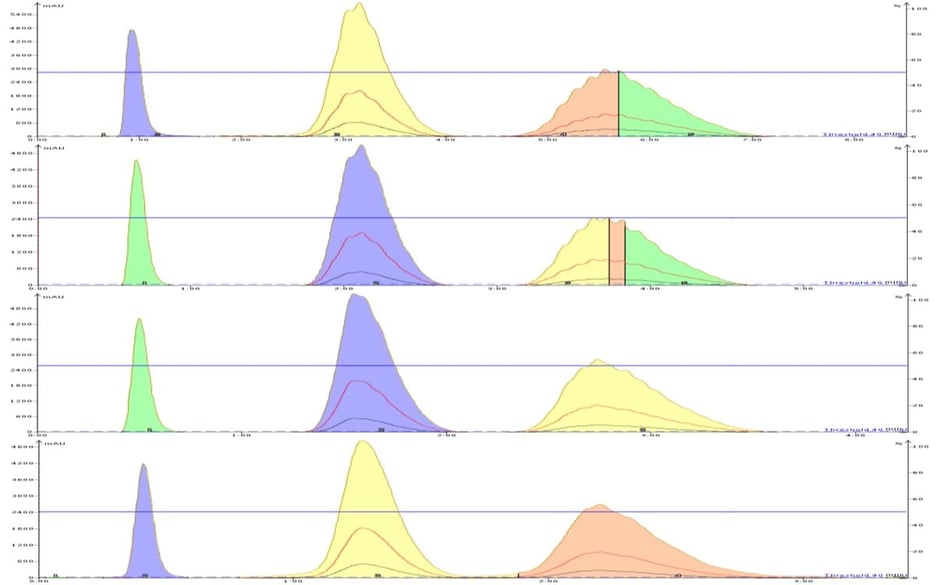 Figure 2. Isocratic separations of two parabens by reversed-phase using a 25 um, 12 gram SNAP Ultra C18 column at 20, 30, 40, and 50 mL/min (top to bottom) show minimal separation decline with increasing flow rate. At 50 mL/min there is some minimal degradation.
Figure 2. Isocratic separations of two parabens by reversed-phase using a 25 um, 12 gram SNAP Ultra C18 column at 20, 30, 40, and 50 mL/min (top to bottom) show minimal separation decline with increasing flow rate. At 50 mL/min there is some minimal degradation.
So, this data says that setting the correct linear velocity is more important with larger particle media than with smaller media and that there is flow rate flexibility in flash chromatography, especially when you use smaller particle columns.
Have you noticed anything different? If so, share your results.
Interested in learning more about flash chromatography? Download the white paper on "Successful Flash Chromatography"!
1 HPLC to UPLC Method Migration: An overview of Key Considerations and Available Tools. Swartz, Michael, Waters Corp., presented at PittCon 2007.
Published: Jan 23, 2023 10:41:52 AM