Jan 18, 2023 11:25:36 AM
What is the purpose of methanol in an extraction method?
By Maura Rury

Have you ever been extracting samples for oil and grease compounds using solid phase extraction (SPE) and thought, “why do I have to use all these different solvents, when I’m just trying to get my compounds to retain on, and then elute from, an SPE disk?”
If you’ve been digging into the extraction method a bit, you’ve probably asked yourself “I wonder what the purpose of the methanol is” at least once or twice. If you’re processing samples for oil and grease, your goal is to determine the concentration of compounds that can be extracted in n-hexane (also known as HEMs), so it’s logical to think that you’d load your sample onto your SPE disk, then pour some hexane through it to elute your target analytes.
For the most part, that logic is sound; however, there’s more to the chemistry than that. I was trying to explain this chemistry to a colleague of mine recently and his eyes started glazing over about 3 minutes into my explanation. I tested my explanation on a few other colleagues and got the same response so I started to give up hope that anyone was going to share my excitement for chemistry and what’s going on within the SPE disk.
Then I stumbled across this graphic and my hope was restored. In this simple graphic, the overall extraction scheme is listed in the center and the addition of methanol and hexane are illustrated to the left and to the right, respectively. What I really like about this graphic is that it allows me to walk through the extraction, step-by-step, and see the impact of each solvent. Let’s walk through it and I’ll show you what I mean.
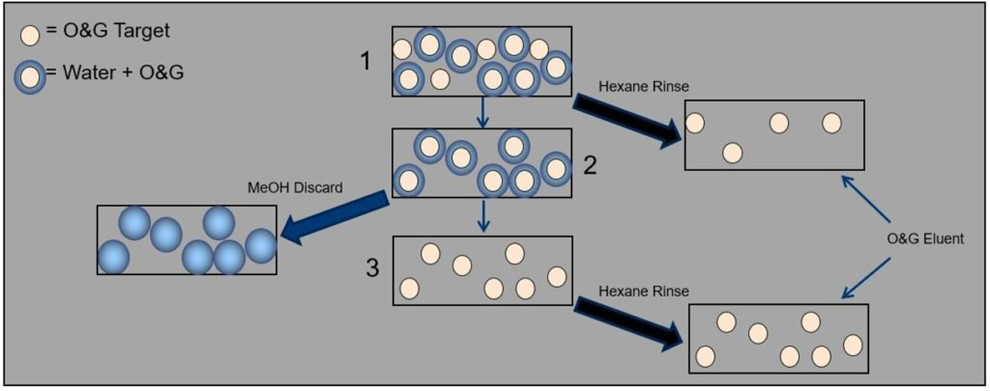
Figure 1: The overall extraction scheme in the center and the addition of methanol and hexane are illustrated to the left and to the right, respectively
If you start at the top, the box labelled “1” shows you the state of your sample after it’s been passed through your SPE disk. The bulk of your water matrix has passed through the disk and been directed to waste, while your target analytes remain trapped in the disk. Some of your target analytes (the beige-coloured circles) are present in solution with water molecules surrounding them. Water molecules are polar and their net dipole moments cause them to be attracted to each other (the positive dipole moment of one water molecule is attracted to the negative dipole moment of another water molecule). So, in their effort to cluster together, water molecules end up trapping a few compounds – compounds that you’d like to extract and quantify. Unfortunately, passing your sample through the SPE disk does not remove these water molecules.
In an ideal world, you would add some hexane, elute all your compounds, then evaporate off the hexane and record your HEM weight. Unfortunately, those pesky water molecules are going to prevent the hexane from reaching some of your compounds (don’t forget that hexane isn’t miscible in water). So you would add hexane to elute the analytes that are free from water molecules and accessible by the hexane (follow the first arrow to the right in the graphic). This will leave you with just the water-bound analytes (the box labelled “2).
Here is where methanol will come to your rescue. Methanol is a polar solvent and is soluble in water. Methanol isn’t as polar as water, but it’s still pretty polar. When methanol passes through your disk, the attraction between methanol molecules and water molecules becomes stronger than the attraction between water molecules and other water molecules. As water molecules seek out methanol molecules, the water molecule clusters break up and release the remaining target analytes you’re trying to extract (i.e. the box labelled “3). One more pass of hexane elutes those target analytes into your collection flask and now you’ve collected all your analytes of interest.
Sceptical of the chemical journey I’ve just outlined? Check out these data tables where the proof is in the numbers. I wanted to see if I could prove out my theory in the lab, so I obtained 6 litres (yes, SIX litres) of a real-world influent sample and divided the sample into six, 1-liter replicates. The six samples were extracted using an automated SPE extraction system. All six of the SPE samples were processed using the exact same system and the same extraction conditions, with one exception – three were extracted using methanol at the appropriate steps and the remaining three were extracted without methanol.
Extraction with methanol
|
Sample ID |
Starting Weight (g) |
Final Weight (g) |
HEM Weight (mg) |
|
Replicate #1 |
6.1831 |
6.2136 |
30.5 |
|
Replicate #2 |
6.2030 |
6.2359 |
32.9 |
|
Replicate #3 |
6.2015 |
6.2357 |
34.2 |
|
Avg. HEM Weight (mg) |
32.5 |
Extraction without methanol
|
Sample ID |
Starting Weight (g) |
Final Weight (g) |
HEM Weight (mg) |
|
Replicate #1 |
6.2473 |
6.2656 |
18.3 |
|
Replicate #2 |
6.1801 |
6.2045 |
24.4 |
|
Replicate #3 |
6.2506 |
6.2643 |
13.7 |
|
|
|
Avg. HEM Weight (mg) |
18.8 |
Under the exact same conditions, the use of methanol produced an HEM weight of 32.5 mg. Without methanol, the average weight of the hexane extractable material was 18.8 mg – a difference of 42% by weight!
Seems like methanol plays a pretty important role in oil and grease extractions!

