Jul 11, 2023 2:00:00 PM
What can I use to purify polar reaction mixtures?
By Bob Bickler

Polar reaction mixtures can be a real challenge to purify with flash chromatography. Traditional normal-phase flash requires toxic dichloromethane (DCM) or chloroform with methanol for these types of purifications which can yield unpredictable, and often un undesirable results. These same compounds are often too polar to be retained on a reversed-phase column and elute at the solvent front (within a single column volume or CV). If you experience one of these issues, a technique called HILIC may be the solution.
So, you may be asking yourself what is HILIC? Well, HILIC is Hydrophilic Interaction LIquid Chromatography. It is a technique used to separate and purify polar compounds that do not retain on reversed-phase and/or are too polar for normal-phase.
Some chemists call this technique reversed reverse-phase chromatography, but actually it is aqueous normal-phase chromatography. Like with normal-phase, HILIC uses a polar stationary phase column (silica, diol, amine, etc.). With HILIC, however, the weaker solvent is typically acetonitrile, while the strong solvent is water. Just so you know, methanol does not usually work for HILIC separations.
Until recently, the only HILIC chromatography I ever performed was in developing a silica flash column QC test (uracil + cytosine in aqueous ammonium acetate) and for sugar separations back in the 80’s when I worked at another company.
Recently, however, I came across a reaction mixture that neither traditional normal phase nor reversed phase could effectively purify, so I tried HILIC. To my delight, this worked well.
The need for HILIC arose when I tried to purify the product of a nicotinuric acid plus benzylamine reaction. While I was able to get some separation by reversed-phase, my product was not resolved well from some by-products making isolation of my target challenging, Figure 1.
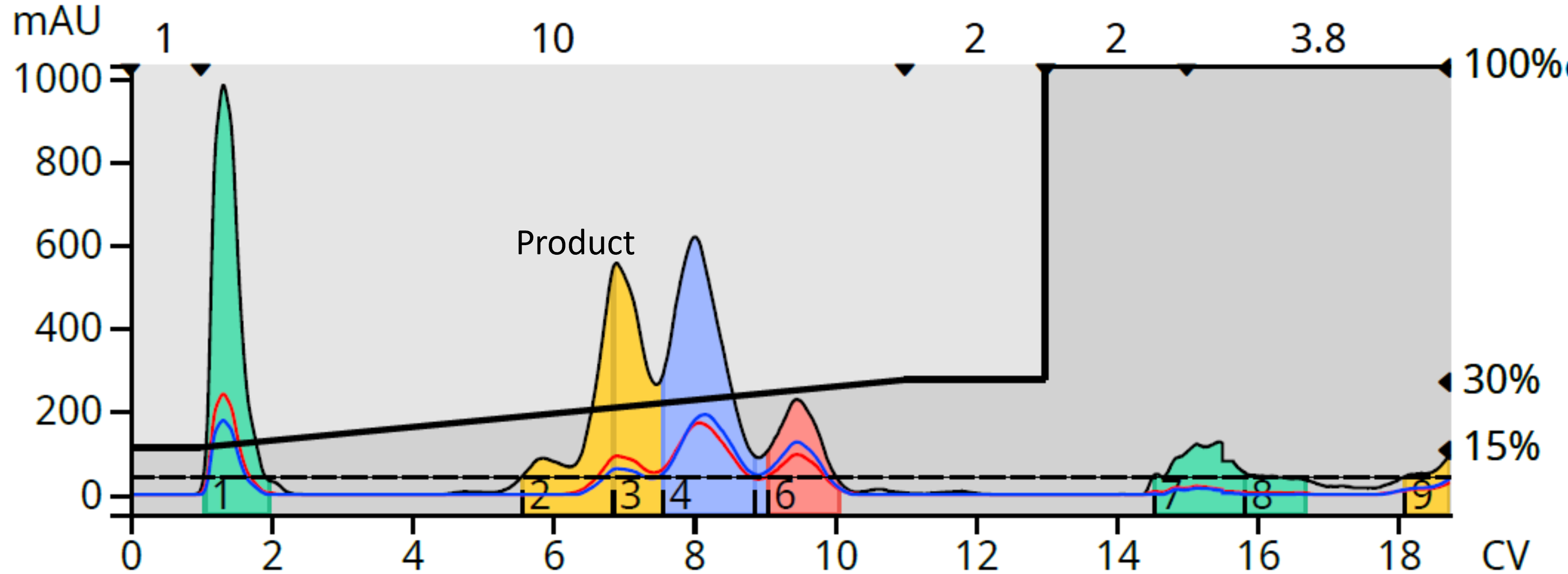
Figure 1. Reversed-phase flash chromatography provided a less than ideal separation.
With the reaction mix only soluble in polar solvents, normal-phase provided limited options. I begrudgingly tried DCM-MeOH (0-30%) with a silica column and achieved a good separation albeit with little room for scaling up the purification, Figure 2.
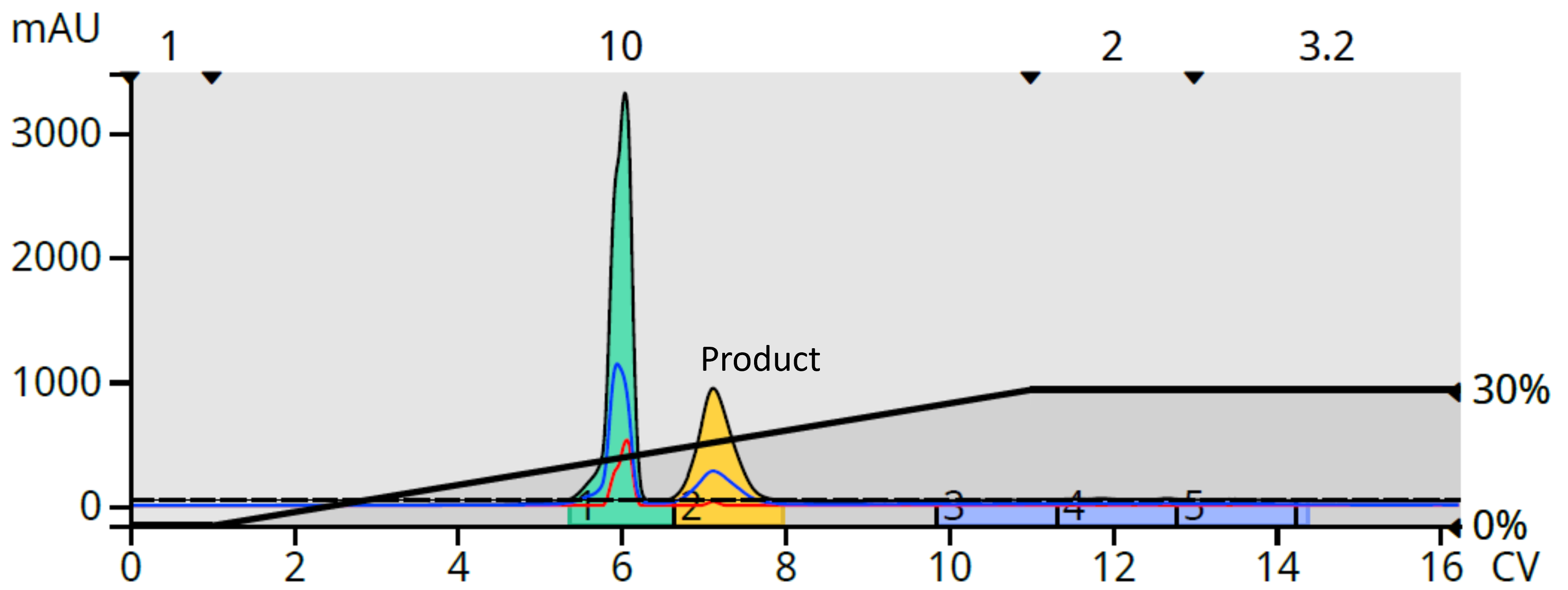
Figure 2. Normal-phase flash chromatography generated a good separation but with marginal resolution.
I then tried HILIC with a silica column (5-gram Biotage® Sfär HC) with a 0-30% water in acetonitrile gradient. This method provided the best results for my needs, fully resolving my product from the by-products, Figure 3.
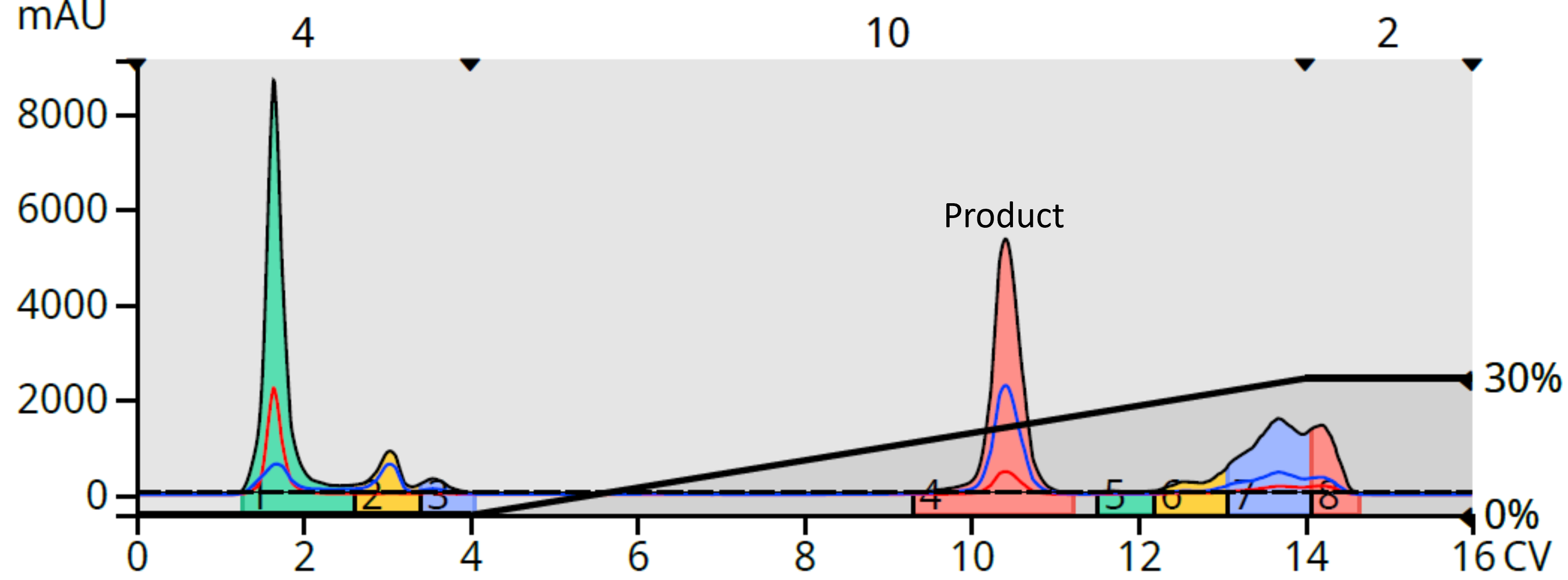
Figure 3. HILIC flash chromatography provided the best separation with excellent resolution between the product and by-products.
HILIC provides advantages over traditional normal phase flash chromatography. Normal-phase is not an environmentally friendly technique using hazardous, toxic organic solvents and single use plastic columns. HILIC, on the other hand, does use acetonitrile, but the strong solvent is water creating a less hazardous waste stream. HILIC also allows silica columns to be reused to an extent (depends on crude reaction mix purity). I have reused silica columns for HILIC applications 4 to 5 times prior to their disposal.
So, if you need to purify polar reaction mixtures, consider using HILIC. It worked for me.
If you are interested in other techniques to make your flash chromatography more sustainable, check out our whitepaper Some Tips for Achieving Greener Flash Chromatography.

