Apr 3, 2023 11:29:30 AM
What is an Isotope Dilution Standard?
By Christopher Mitchell

When it comes to chromatographic analysis, it’s no surprise that the analytes of interest are the main focus. After all, those are the compounds one wishes to isolate and quantitate to report to their clients. The analyzer of choice can vary from lab to lab, but one thing will remain true no matter the method; internal standardization is required. By adding an internal standard to your samples you’re helping to correct any normal analyte loss that will be experienced in sample prep or analysis. As simple as this sounds your method can become quite problematic if the wrong internal standardization isn’t employed. While we can explore the various ways to standardize your samples, I’ll focus on one that is quite important.
Isotope Dilutions
Before we break down isotope dilutions, let’s first discuss what an isotope is. An isotope is a distinct nuclear species of the same element. They contain the same number of protons but differ in their neutron amounts in their nuclei. For Instance, carbon-12 and carbon-13 are two isotopes of the carbon atom. Both have atomic numbers of 6 but their mass numbers, 12 and 13 respectively, differ due to their neutron counts. Isotopes can be used to determine the concentration of substances by spiking known concentrations directly to a pre-extracted sample and then using the ratio of the peak area of the native analyte to that of an isotopically labeled analogue. This is known as the isotope dilution method. The reason why Isotope dilutions methods work well for LC-MS/MS analysis is that isotopes have nearly identical chemical properties which allow them similar to separate chromatographically. Once separated, it is easy to then compare the signal ratio vs intensity, which is why isotope dilution is considered one of the most precise methods for measuring analyte concentrations.
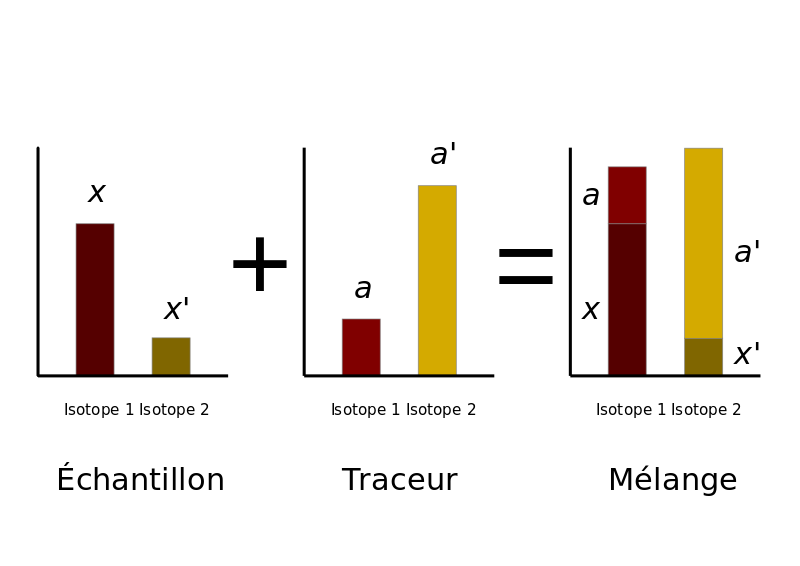 Reference: https://commons.wikimedia.org/wiki/File:Sch%C3%A9ma_dilution_isotopique_2015.svg
Reference: https://commons.wikimedia.org/wiki/File:Sch%C3%A9ma_dilution_isotopique_2015.svg
The figure above shows isotope dilution in its simplest form visually. When you take your known spike concentrations and add them to the sample the resulting mix will contain both an unknown sample concentration along with the known sample spike concentration and quantitation is then calculated by the comparing ratios.
To use or not to use
It’s already been discussed how Isotope Dilution is a truly reliable method when compared to other methods of internal standardization. Another advantage is after spike and sample have been mixed, loss in substance no longer has an effect on the result of the analysis so it is extremely ideal when dealing with low-recovery analytes. That doesn’t necessarily mean it isn’t without its limitations. For some monoisotopic elements isotope dilution won’t be able to be employed since those compounds only have one stable isotope. In addition, it can be somewhat destructive as it requires dissolution of the sample to homogenize with the spike. In the end, it is up to the laboratory to decide what technique to utilize for internal standardization of their analytical procedures. In addition, laboratories may need to adhere to regulatory guidelines and method criteria. A great example of this is EPA method 533 which requires Isotope Dilution for quantification of Per- and Polyfluorinated Substances (PFAS) in drinking water.
Another advantage is after spike and sample have been mixed, loss in substance no longer has an effect on the result of the analysis so it is extremely ideal when dealing with low-recovery analytes. That doesn’t necessarily mean it isn’t without its limitations. For some monoisotopic elements isotope dilution won’t be able to be employed since those compounds only have one stable isotope. In addition, it can be somewhat destructive as it requires dissolution of the sample to homogenize with the spike. In the end, it is up to the laboratory to decide what technique to utilize for internal standardization of their analytical procedures. In addition, laboratories may need to adhere to regulatory guidelines and method criteria. A great example of this is EPA method 533 which requires Isotope Dilution for quantification of Per- and Polyfluorinated Substances (PFAS) in drinking water.
For more information on Isotope Dilutions follow the link below!

