Feb 10, 2023 3:18:56 PM
Which loading method should I use for purification by flash chromatography?
By Bob Bickler

Getting the most benefit from your crude sample purification with column chromatography or flash chromatography involves optimizing many variables. In previous posts I have talked about selecting the best solvents, their ratios, and maximizing load based on TLC Rf data. These are all important chromatography-generated variables but now I would like to share some tips on actual technique differences and their impact on purification performance.
In particular in this post I will focus on the benefits and drawbacks of liquid loading and dry loading. Both have their place in liquid chromatography but when should one technique be used over another?
You have performed a synthesis, let’s say in a polar solvent like acetonitrile, and you need to use flash chromatography to purify your product. You performed normal-phase TLC which gives a suitable separation using hexanes/ethyl acetate and now you are ready to purify the mix by flash chromatography – do you inject the reaction mix directly onto the cartridge?
I would not because of acetonitrile’s polarity. If your injection/dissolution solvent is more polar than the gradient initial solvent ratio, your chances of getting good a separation/purification will typically be reduced. The reason for this is that your synthesized product and by-products are likely more lipophilic than the polar dissolution solvent meaning their adsorption onto silica will be inhibited by acetonitrile, which is in greater volume than the reaction products and therefore is preferentially adsorbed onto silica. In some instances very lipophilic compounds will still chromatograph as desired because they do not adsorb to a great degree anyway. You can try injecting a polar solution but in order to get the quality separation you need a very small injection volume.
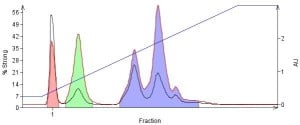
As an example, 0.1 mL of a methanol solution was injected on a 10 g silica cartridge and separated using a hexane/ethyl acetate gradient, Figure 1. The injection volume was less than 1% of the column volume and still caused major separation problems for the more polar/well retained compounds.
Figure 1. Normal-phase separation of a crude mix dissolved in methanol shows the impact of a polar injection solvent on a separation, even with a volume <1% of the column volume. Because the polar solvent is in high concentration it is adsorbed preferentially and deactivates the silica. Poorly retained compounds are often not impacted at the same level as more polar components.
Another option is to perform a dry load. With dry loading you blend your dissolved mix with an adsorbent – silica, diatomaceous earth, Florisil®, etc. and allow the solvent to evaporate until the mix is a free flowing powder. This dry compound-bearing media is then transferred to an empty vessel attached to the inlet of the flash cartridge, Figure 2. By evaporating the dissolution solvent its impact on compound adsorption and subsequent separation is eliminated resulting in a vastly improved separation, Figure 3.

Figure 2. Dry loading vessel (DLV) commonly used to improve purification of sample mixtures dissolved in volatile polar solvents.
A potential down side to dry loading by this method is sample dilution, not with solvent but with sorbent. This occurs if too much sorbent is used for the amount of reaction mixture you need to purify because you are spreading the reaction mixture throughout whatever amount of sorbent you have used. If too much sorbent is used broader elution bands than what is optimal are generated during elution, even with gradient elution. This issue lessens when purely absorptive materials like diatomaceous earth are used. For optimal dry loading with silica, I use a ratio of no more than 1:3 crude mix to sorbent (1 gram of reaction mix:3 grams of sorbent); 1:2 also works very well.
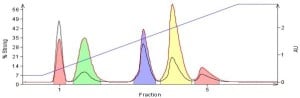
Figure 3. Dry load chromatography of the 5-component mix. The crude mix was blended with silica at a ratio of 1:3 and the polar solvent evaporated. This loading technique eliminates the dissolution solvent's impact on the separation and yields superior purification results compared to liquid loading.
A variation to this "external" dry load technique is to add your crude mix to a smaller, pre-packed cartridge of either the same media as the purification column or packed with another sorbent as mentioned above. Once loaded onto the top of the sorbent the solvent can be evaporated by pulling a vacuum through the cartridge. This technique concentrates the sample on the sorbent in a tighter band than the above mentioned technique, which will improve the separation quality.
As examples, compare Figures 3 and 4. In Figure 3, an external dry load method was used as an alternative to a liquid load (Figure 1). The external dry load method dramatically improves the separation compared to the liquid load but causes some band-broadening due to the spreading of the sample over the total amount of dry load sorbent. However, by using an internal dry loading vessel as discussed above and concentrating the sample on less silica the elution bands or peaks remain sharp reducing collection volume and therefore fraction evaporation t
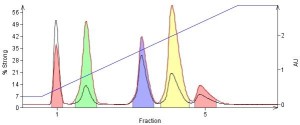
Figure 4. Dry loading with a pre-packed Samplet® cartridge (Biotage) shows further separation improvement (sharper peaks with better resolution) compared to both liquid and pre-blended dry loading.
Liquid injections are simpler and faster than dry loading but the impact of the dissolution solvent must be considered - both its chemistry and injection volume. In my work I use liquid loading if I can concentrate my mix in a minimal amount of a moderately polar solvent such as DCM or acetone. If I can get a 1g/mL concentration, then I can inject a small volume of the mix to achieve my purification goal. If getting the mix dissolved at near that concentration is not possible, I will use dry loading.
Interested in learning more about flash chromatography? Click the link below to watch my webinar Inspiring Productivity with Modern Flash Chromatography