Jan 24, 2023 3:25:57 PM
What is the maximum flash column sample load volume?
By Bob Bickler

A fairly common question I receive is "how much of my reaction mixture can I load on my flash chromatography column?". To this question there is not a cut and dry answer because of a number of variables which influence the outcome. To help illustrate this, I will show in this post the results of a study I performed in my lab with one of my reaction mixtures.
So, I mentioned that there are several variables involved in determining column loading capacity. These include…
- Reaction mixture solubility in both the mobile phase and the dissolution solvent
- Loading volume (% of the column’s internal volume)
- Loading mass (% of the column’s media weight)
- Dissolution solvent polarity
- Column size
- %B solvent in the gradient
For this post my reaction mixture was dissolved in acetone, a medium polarity solvent. Higher polarity solvents would also work but because solvent polarity dramatically impacts purification load volume (higher polarity = lower volume), I used about the lowest polarity solvent possible.
Because flash chromatography is performed using various column sizes, specifying an injection volume maximum for each column size is a bit impractical so it makes more sense to have load volume determined as a percentage of a column volume (CV). Using loading as % of CV provides you with a better guideline for any column size you may use.
For this study, I used a 10-gram Biotage® Sfär HC silica column and a 7-60% EtOAc in hexanes linear gradient over 10 CV. My 10-gram column had a CV of 15 mL (listed on its label), Figure 1.
-Dec-12-2022-01-33-10-1155-PM.jpg?width=3024&name=10-g%20Sfar%20label%20(2)-Dec-12-2022-01-33-10-1155-PM.jpg) Figure 1. 10-g Biotage®Sfär label showing its column volume (CV) and other important information.
Figure 1. 10-g Biotage®Sfär label showing its column volume (CV) and other important information.
My reaction mixture (343 mg) was dissolved in 5 mL acetone for a concentration of 68.6 mg/mL. I loaded increasingly larger reaction mixture volumes, from 0.1 mL (0.67% CV) to 2 mL (13.33% CV) to see the impact they had on the results, Figure 2.
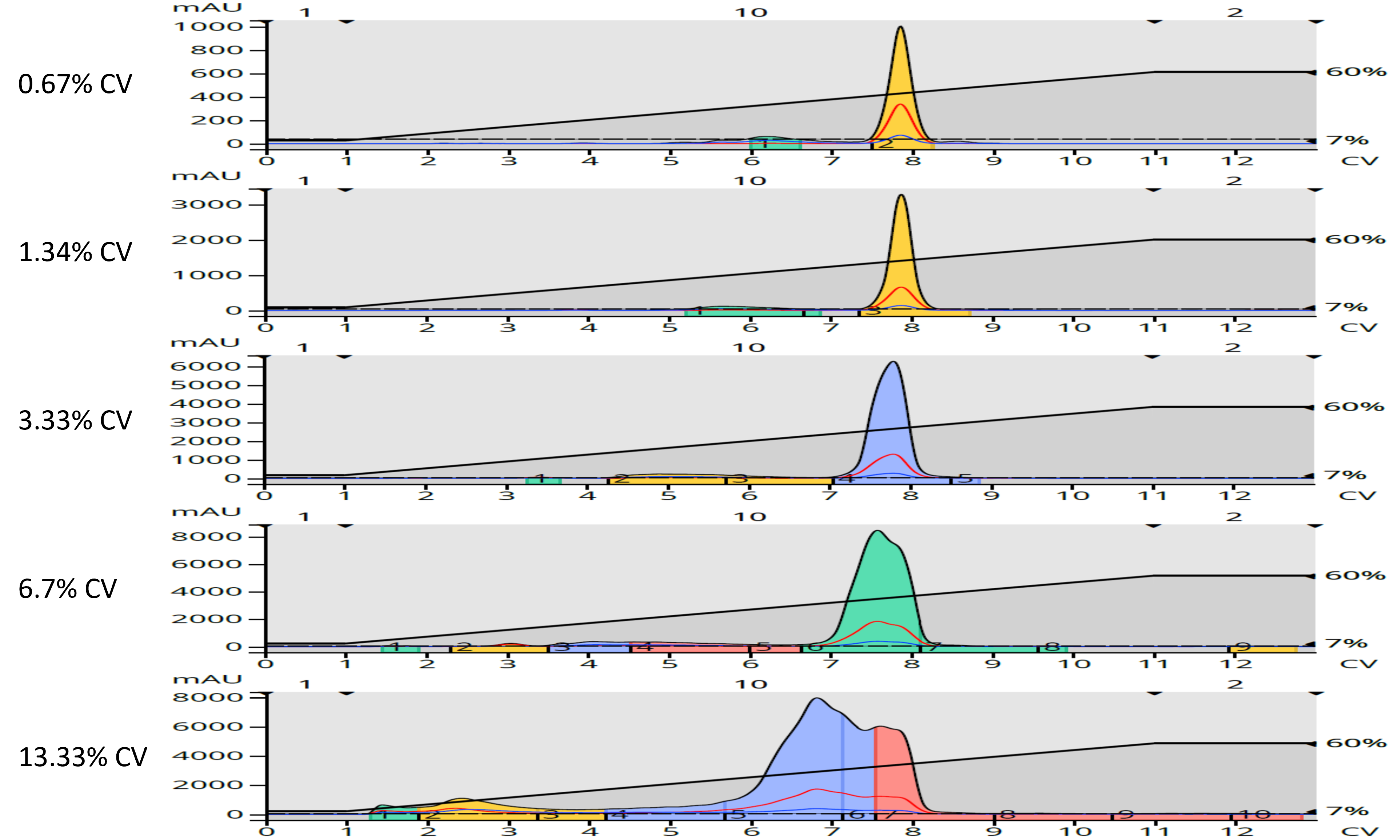 Figure 2. Normal-phase load volume impact for a reaction mixture dissolved in acetone and purified on a 10-g silica column. As injection volume increased, the compounds (peaks) broadened and were displaced causing earlier elution.
Figure 2. Normal-phase load volume impact for a reaction mixture dissolved in acetone and purified on a 10-g silica column. As injection volume increased, the compounds (peaks) broadened and were displaced causing earlier elution.
The results show that with increasing injection volume there is a displacement effect that happens. The injection solvent, whatever volume it may be, displaces a corresponding and equal volume in the column when it is injected. If the injection solvent is more polar than the mobile phase, which is usually the case, then the sample’s components are impeded from adsorbing to the silica by the injection solvent and elute earlier than with lower injection volumes, which is easily seen in Figure 2. If the volume is large enough, overall mass transfer is impacted giving rise to elution band splitting (peak splitting), also evident, especially with the 1 mL (6.7%) and 2 mL (13.33% CV) loads in Figure 2.
So, based on these results, I would not load more than 3% of a column’s internal volume (CV) as larger injection volumes will degrade the separation and reduce target compound purity.
For more information of flash chromatography, please click here.

