Jan 19, 2023 7:17:59 PM
When should I use an amine-bonded silica for flash chromatography?
By Bob Bickler

Flash chromatography is a standard part of an organic chemist’s workflow. It is utilized after most reaction steps in order to remove most of the generated by-products and excess reagents.
For 90% of all flash chromatography applications, chemists use unbonded silica. For the other 10%, alternative media is utilized including reversed-phase (C18), amine-bonded silica (NH2), or some other sorbent.
In this forum I have frequently discussed using silica and reversed-phase but have spent little time talking about amine-bonded or amine-functionalized silica and why and where it is best suited.
Let’s focus on the chemistry of the different media.
Silica
Silica is typically synthetic, made from sodium silicate and the reaction conditions designed to provide specific properties such as surface area, porosity, and particle size. Silica is available in both spherical and granular (irregular) particle shapes. Spherical silica is made to a specific set of parameters as mentioned previously. Granular silica is also made to specific specifications but with a large particle diameter. This silica is then ground down to create smaller particles which are then separated into various particle size or mesh ranges.
Silica contains various silanol (Si-OH) functional groups in different ratios. Though chromatographic silica is typically pH neutral, the silanols are acidic (Brønsted acids) and can cause issues when trying to purify organic bases – the basic compounds stick to the acidic silica and are then difficult to remove and separate. In cases like this, solvents with greater polarity, e.g. methanol with an added base can be useful to get the desired results. This, however, such purification methods can be time consuming to develop and do not always work.
Reversed-phase
Reversed-phase media is silica bonded with a hydrophobic reagent (e.g. octadecyl silane). This media, if well bonded, allows organic bases to separate and elute using water-based solvent systems. However, to reduce elution band width and improve resolution, pH modifiers or buffers are commonly used and even then, the results may not be as good as you need.
Amine-bonded
Amine-bonded silica provides chemists an alternative to silica columns and base-modified organic solvents as well as reversed-phase columns also using buffered or pH-adjusted aqueous solvent systems.
The amine-bonded silica has the pH modifier attached to its surface which decreases the underlying silica’s acidity and creates a chemical environment more conducive to basic compound chromatography as seen in Figure 1.
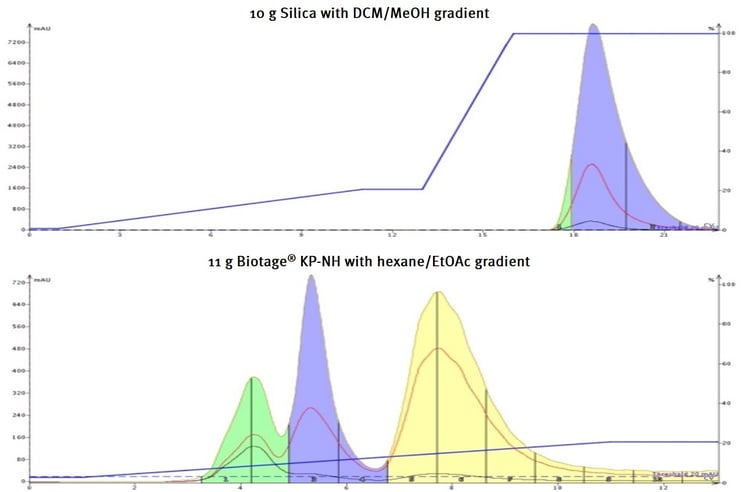
Figure 1. Flash chromatographic separation of three basic tricyclic amines (all with pKa >9). Top - silica column with a DCM-MeOH gradient failed to separate the compounds and required 100% methanol to elute. Bottom - amine-bonded silica column elutes and separates the three compounds using a hexane/ethyl acetate gradient.
In this case, a mixture of three tricyclic amines (all strongly basic) were injected onto a silica column and an amine-bonded silica column (Biotage® Sfär KP-Amino). The compounds were separated first on a silica TLC plate and a KP-Amine TLC plate using different solvents.
Silica – 80% DCM + 20% MeOH
KP-NH – 90% hexane + 10% ethyl acetate
Though the compounds all separate on both plates (data not shown), the compounds do not elute from the silica column until it is flushed with 100% methanol – without separation. The KP-amine column, however, easily separates and elutes the three basic tricyclic amines with just a mix of hexane and ethyl acetate.
Amine-bonded silica is actually quite versatile in its application range. Because the amine functionality is pi-electron rich it is useful in purifying other pi-electron rich compounds.
In one such situation, a prospective customer from Randolph-Macon College [1] synthesized a porphyrin compound, meso-tetra-(p-bromophenyl) porphyrin [2], Figure 2, and asked for our assistance in its purification.
/Blogs%20ONLY/Tetra(p-bromophenyl)porphyrin_300.png?width=300&height=300&name=Tetra(p-bromophenyl)porphyrin_300.png)
Figure 2. Meso-tetra-(p-bromophenyl) porphyrin structure.
I tried both silica and reversed-phase columns to purify the reaction mixture. No separation was achieved using reversed-phase but normal-phase silica with a hexane-ethyl acetate gradient provided a good, but not great, separation, Figure 3.
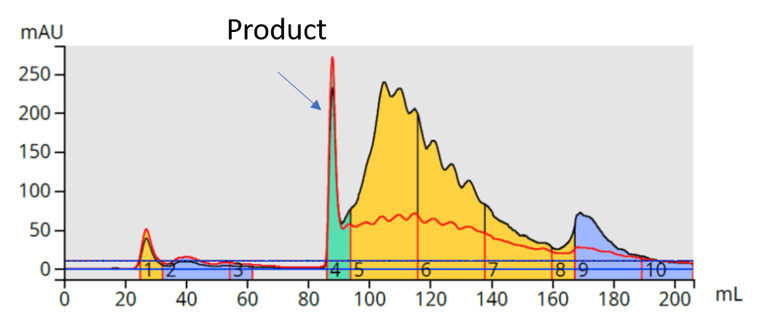
Figure 3. Silica column purification of meso-tetra-(p-bromophenyl) porphyrin reaction mixture using a hexane-ethyl acetate gradient partially separates the product from its reaction by-product impurities.
Not satisfied with the results, I then tried a Biotage® Sfär Amino column, also using a hexane-ethyl acetate gradient, and this combination worked exceedingly well [3], Figure 4.
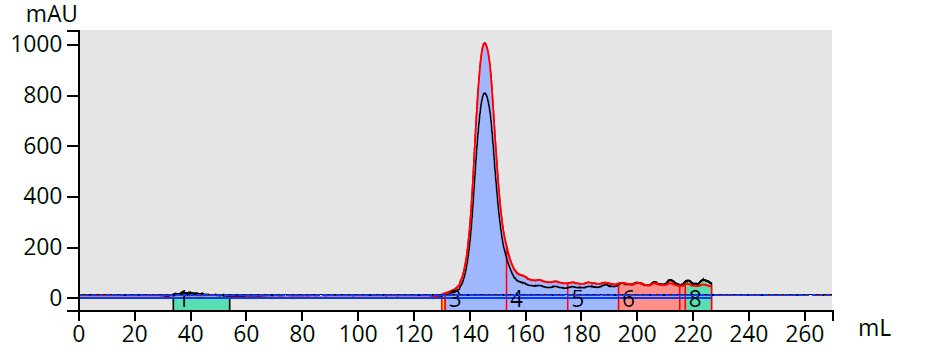
Figure 4. Amine-bonded column purification of meso-tetra-(p-bromophenyl) porphyrin reaction mixture fully separates the product from its impurities, which did not elute.
So, why did the amine-bonded column work best? The porphyrin structure contains four amines which are attracted to silica’s acidic silanols. Both the product and its by-products apparently have affinity for the silica making their separation possible, but incomplete. The amine column, however, selectively binds the by-products allowing the product to separate from them and elute as a pure, purple fraction.
What we have learned with these examples is if your compounds are organic and contain pi-electron functional groups, an amine-bonded flash column should be tried first rather than experimenting with silica or even reversed-phase chromatography.
If you are interested in learning more about flash chromatography, download our whitepaper – Successful Flash Chromatography.
[1] Sample courtesy of John Thoburn, PhD. of Randolph-Macon College
[2] https://pubchem.ncbi.nlm.nih.gov/compound/Tetra_p-bromophenyl_porphyrin
[3] Data used with written permission from John Thoburn, PhD., Randolph-Macon College
Published: Jan 19, 2023 7:17:59 PM

