Feb 6, 2023 12:05:07 PM
Does dry load media choice impact reversed-phase flash purification results?
By Bob Bickler

Flash chromatography is a purification technique used by chemists to isolate their targeted compound from by-products and impurities. Because the reaction mixture (or natural product extract) may have its best solubility in a solvent that is chromatographically “stronger” than the mobile phase, liquid sample loading can be problematic causing early eluting and/or broad peaks with poor purity. In those cases, a technique called dry loading is frequently used.
With dry loading, the extract or reaction mixture is blended with either a non-interactive sorbent (silica, diatomaceous earth, C18, alumina), or with an interactive media such as an ion exchanger.
I know this is a lot of dry load media options. How do you know which to choose and what impact will the chosen media have on you purification results?
In this post, I report on some chromatography research I conducted on a microwave synthesis reaction (Biotage® Initiator+), Figure 1, where dry loading with five media was compared to a liquid load to determine what, if any, improvements were created.
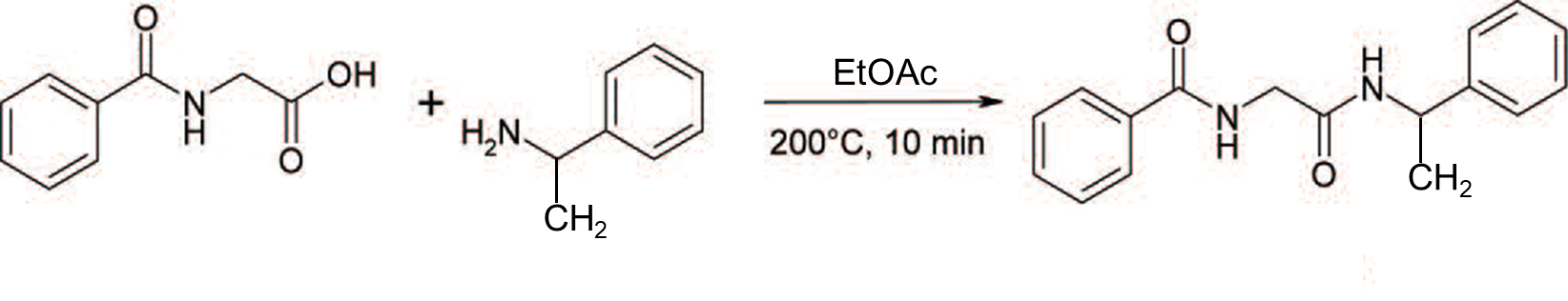
Figure 1. Reaction used to create test mixture.
As I mentioned above, the goal of the research was to determine what, if any, influence a dry load sorbent could play in the purification of a reaction mixture. For this work I selected the following dry load sorbents offered by Biotage which differ in functionality.
- Non-interactive/inert
- Biotage® KP-C18-HS
- Biotage® HM-N-R (a diatomaceous earth media)
- Biotage® KP-Sil
- Interactive
- ISOLUTE® NH2 (anion exchange)
- ISOLUTE® SCX-2 (cation exchange)
The synthetic product and by-products from this reaction were too polar for normal-phase chromatography with hexane/ethyl acetate so I chose to use reversed-phase instead. To establish a “baseline” for comparison, I first generated data using a liquid load (0.5 mL) and a 12-gram Biotage® Sfär C18 column. For this work I used a Biotage® Isolera Dalton 2000 to better track changes in the chromatography results.
Liquid Loading
The liquid load purification (93 mg) completely separated three compounds with the synthetic target eluting last, Figure 2. Each compound was both UV and mass detectable.
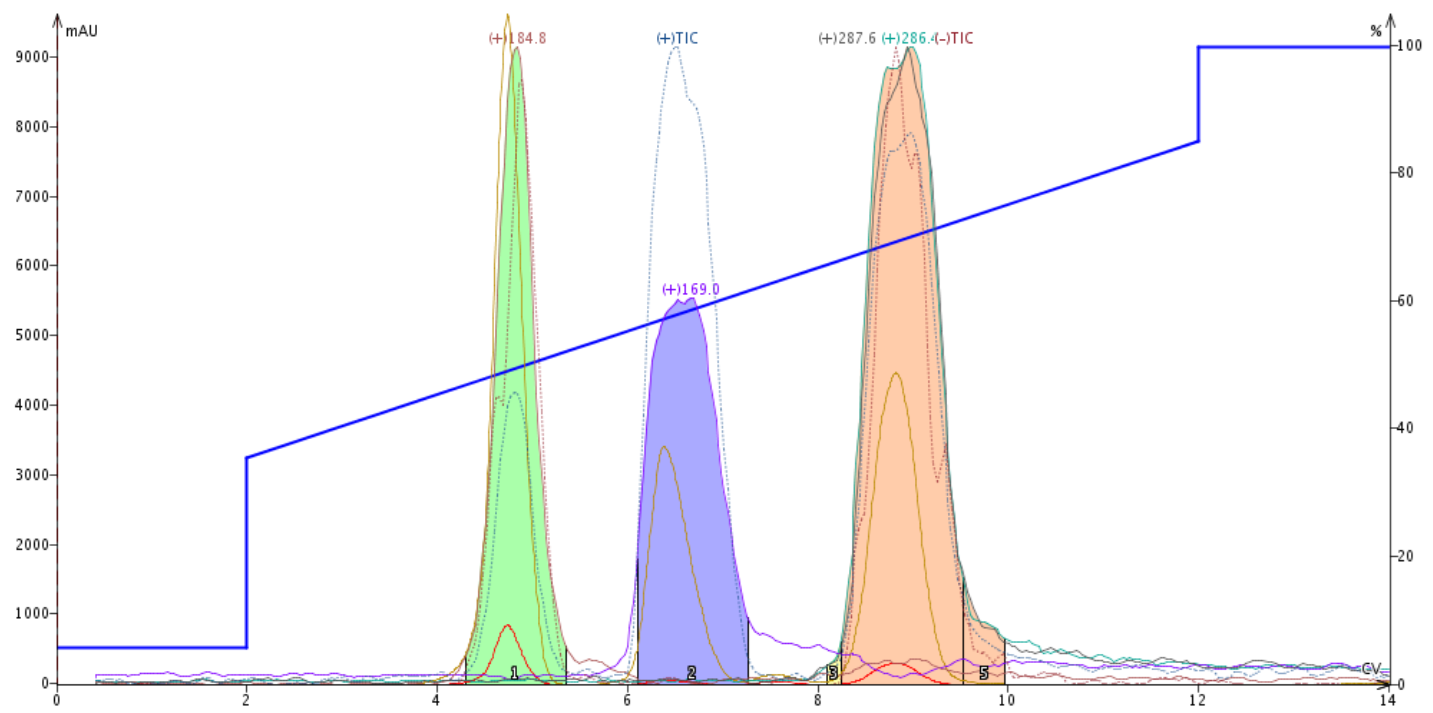
Figure 2. Reaction mixture purification using liquid loading (0.5 mL) provides an excellent separation of the product (pink) and its two major, more polar by-products.
Dry Loading
For dry loading I mixed 0.5 mL of the reaction mixture (93 mg) with 0.3 grams of each sorbent and dried them using a Biotage® V-10 Touch rapid solvent evaporator. Chromatographically, the three non-interactive media provided similar results to the liquid load with the major changes being a marked narrowing of the elution bands (peak widths) which improves compound resolution.
These results are typical when dry loading because there is more available media to perform the separation (the dry load media increases the purification media volume and surface area). Also, by drying the reaction mix onto the dry load media, the band-broadening effects of liquid loading are eliminated generating tighter elution bands, Figure 3.
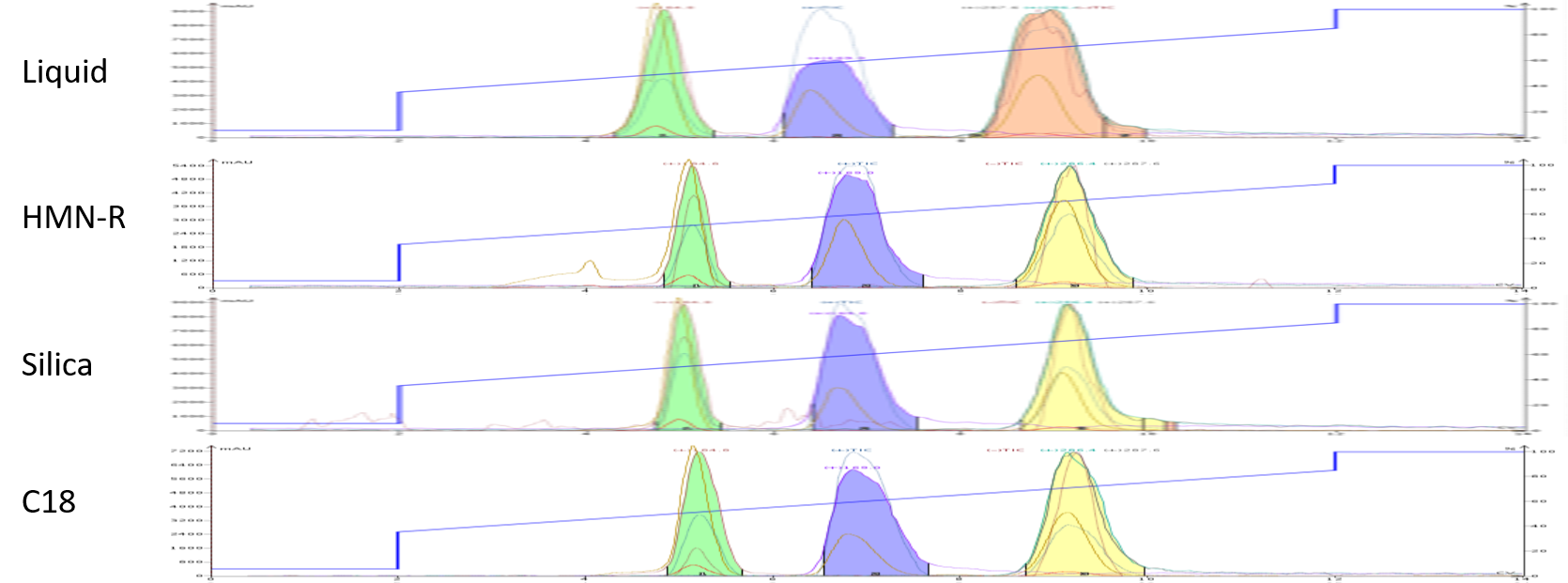
Figure 3. Comparison of liquid loading and dry loading techniques for a reaction mixture purification. Dry loading with inert media provided tighter elution bands and improved peak resolution compared with liquid loading.
The two potentially interactive media, NH2 and SCX-2, provided different results. While the SCX-2 media was inert towards the sample components generating chromatography matching the inert media, the NH2 media scavenged most of the first eluting compound, an indication it was an anionic by-product, Figure 4.

Figure 4. Dry load purification of the reaction mixture using functionalized media. Top - ISOLUTE NH2 media scavenged most of the first eluting by-product. Bottom - ISOLUTE SCX-2 media provided results similar to the inert dry load media.
So, what do these results tell us? To start, knowing your sample’s chemistry is helpful. If you know your product is not likely ionizable, acidic, or basic in solution then any dry load media will work. If indeed you have an interfering by-product of unknown chemistry, try an ion exchange dry load media as it may greatly enhance your target compound’s purity.
More information on dry loading can be found at this next blog!

