Feb 7, 2023 10:38:49 AM
High-throughput Protein Purification Strategies
By Biotage

Antibodies are indispensable tools that are used in a variety of research applications, diagnostics, and therapeutics. Often, these applications require purified antibodies which means that the antibodies must be isolated from a biochemical mixture such as serum or cell culture supernatant. As a result, antibody purification is an important procedure that has implications for multiple downstream uses.Efficient antibody purification can also improve the stability of these molecules which is useful for long-term storage.
Several techniques are available for antibody purification, but affinity chromatography is the most commonly used. One of the main benefits of affinity purification is the high selectivity obtained which enables opportunities to avoid multistep purifications in research and drug discovery applications.
Traditional small-scale chromatography purification formats for antibodies and other biomolecules include magnetic beads, spin columns, miniaturized columns, and filter plates. However, all these technologies suffer from different drawbacks in enabling efficient high-throughput purification. For example, magnetic beads are highly selective but often suffer from lower capacity and the requirements for specific and expensive instrumentation, whereas spin columns are hard to automate and sample dilution and reproducibility can be a challenge. Likewise, filter well plates offer complex automation protocols and eluted samples are often highly diluted. Miniaturized columns are successfully and most commonly used for early process development. They offer scale-down models of purification steps but are limited to 8 samples/run. However, these miniaturized columns have their sweet spots at slightly larger resin volumes than the other formats mentioned and are less efficient for high throughput purification.
Overview of small-scale chromatography purification formats
Table 1 shows a high-level comparison of some of the key technical parameters of importance for small-scale biomolecule purification in research and development. Figures and numbers are claimed from respective product specifications and/or available protocol recommendations from suppliers. The formats have different pros and cons depending on what scale, throughput, and concentration is required.
| PhyTip Columns | Filter plates | Miniaturized Columns | Magnetic beads |
Dispersive solid-phase tip purification |
Spin columns | |
| Speed | ~ 15 min | >60 min | >120 min | >60 min | 10-30 min | >60 min |
| Resin scale | 5-160 µL | 6-50 µL | 50-600 µL | ** | 5-100 µL | 5-100 µL |
| mAb capacity (estimated)* | ~1.1 mg | ~1.5 mg | ~1.2mg | < 0.05 mg** | ~1.0 mg | ~0.9 mg |
| Elution volume* | 120 µL | 3 x 200 µL | 3x50 uL | 100 µL | ~400 µL | 3 x 50 µL |
* Shown for Protein A resins, 50 uL resin - 40 uL resin for the PhyTip format
** Depends on the volume of mag beads used - this example assumes 50 uL beads slurry for 1 mL sample
Table 1: Comparison of key features of different small-scale purification formats. The table compares key characteristics at a 50 ul resin scale (40 ul for PhyTip columns as the 50 ul resin volume is not available in this format)
PhyTip columns
PhyTip® columns are an attractive alternative to traditional purification formats and procedures. The columns are composed of a chromatographic resin packed between two thin screens in the tip of a single-use pipette tip (Fig 1 below). The dual-flow chromatography technology is used to produce consistent yields at high recovery in small elution volumes due to the packed resin bed. Minimizing the amount of purified sample that is obtained by high throughput purification methods allows for the most efficient use of resources, as screening and selection approaches often aspire to test thousands of antibody variants in parallel. Small elution volumes are also often desirable to achieve the highest possible concentration of the protein of interest since sensitivity and detection limits are frequently limiting factors in the downstream analyses.
One of the major advantages of this technology is its automation friendliness. They are designed to operate on all major liquid-handling robot platforms including Hamilton, Tecan, Dynamic Devices, Beckman, Opentrons, Analytik Jena, Perkin Elmer, and Agilent Technologies. There is low overhead and no additional equipment needed for compatibility, making it an ideal alternative for high throughput purification. The run time for a full purification protocol of 96 samples can be as short as 15 minutes depending on sample volume. The methods are easy to develop, and validated protocols are available for use with many of these platforms.
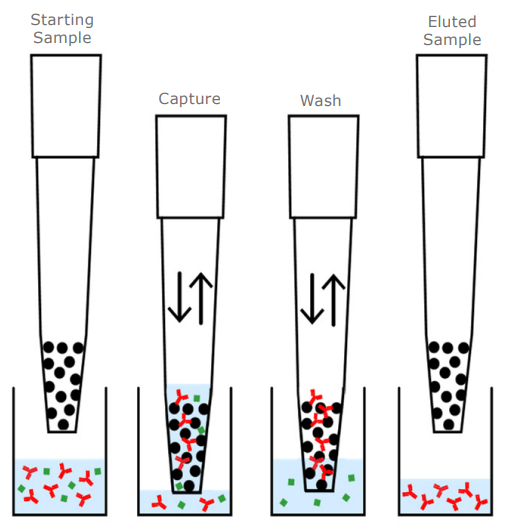 |
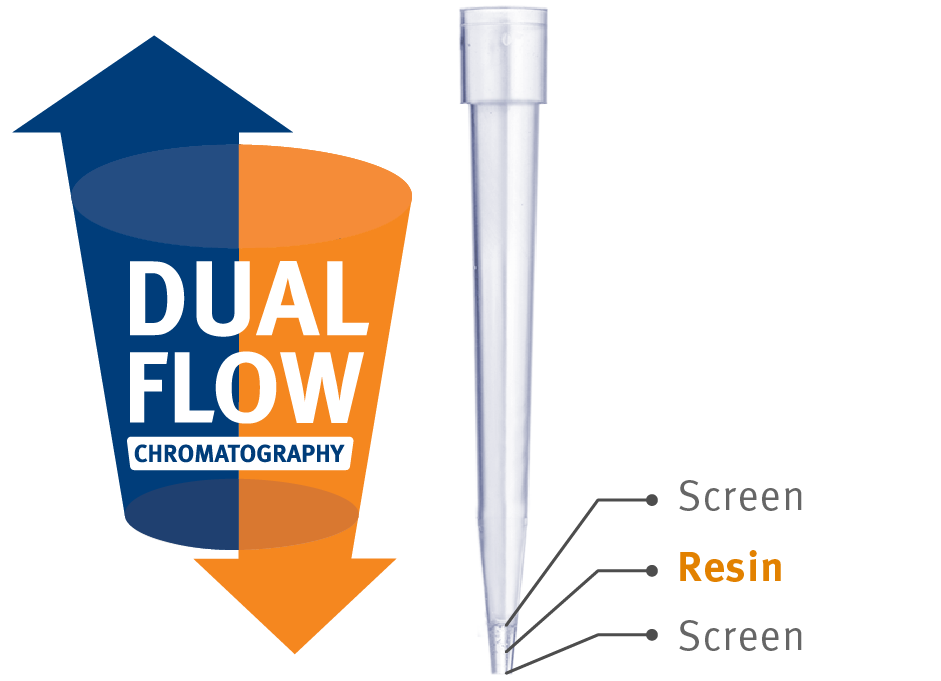 |
Figure 1. Principles of dual flow chromatography in PhyTip columns.
The unique column design with a packed resin bed binds the entire sample to a small resin bed volume. Basic chromatographic principals control the separations, and the bi-directional flow drives interactions to high-loading equilibrium regardless of kinetic rate constants. The purified sample can be eluted in small volumes, resulting in a highly concentrated sample. Dual-flow chromatography is a gentle purification process producing high levels of biologically active protein
Case study – Sample concentration
One of the key advantages of PhyTip column technology is the small elution volume. This is of key importance as the highest possible concentration of the protein of interest is critical for sensitivity and detection limits in the following analysis. Figure 2 below shows how PhyTip column technology outperformed spin columns and magnetic beads technology with regard to yield and concentration of a purified HIS-tagged sample (Courtesy of Vertex Pharmaceuticals). Figure 3 shows how the very low resin volumes in PhyTip columns enable high recovery and high concentration of the eluted sample.
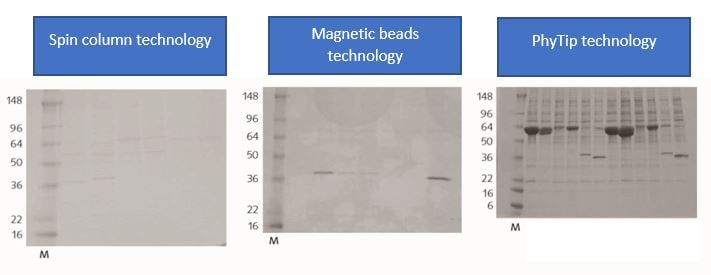
Figure 2. Comparison of concentration effect of spin-column technology, magnetic bead technology, and PhyTip column technology

Figure 3. Purification of low concentration of IgG using spin columns and different resin volumes in PhyTip columns.
Conclusions
PhyTip columns enable you to automate the small-scale protein purification process and offer several advantages over traditional small-scale chromatography methods such as:
• High Throughput
• Consistent high yields and concentrations
• Complete walkaway automation
• Ease of implementation
Compared to traditional small-scale protein purification technologies such as magnetic beads and spin columns, PhyTip columns offer easier automation and higher concentrations of purified sample. This enables optimal sample preparation for the analytical detection and characterization of the purified samples which is often a limiting factor.
They also work with all major robotic platforms and come as prepacked columns that are ready to use. This means that you can use existing platforms that are already available in your lab, making PhyTip column technology easy to implement and use. And another great feature? These columns not only work with antibodies but can also be used to purify other proteins, viral vectors, and plasmids.
For more information, check out our PhyTip columns brochure or take a look at our webinar available here, or contact one of our support teams who can help answer any questions and get you started on your PhyTip journey today.


